推薦產品
化驗
97%
bp
285 °C (lit.)
mp
52-55 °C (lit.)
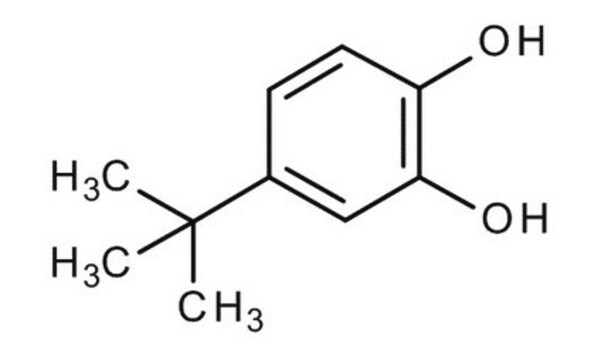
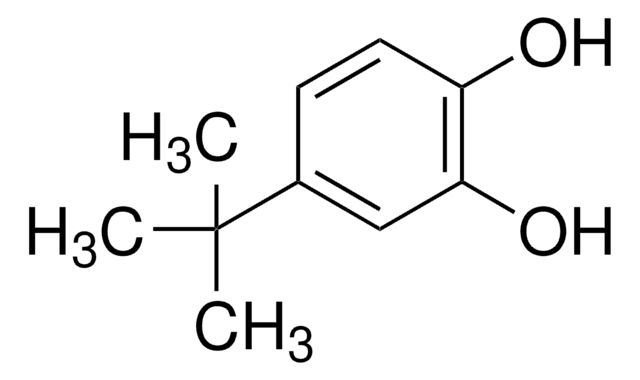
SMILES 字串
CC(C)(C)c1ccc(O)c(O)c1
InChI
1S/C10H14O2/c1-10(2,3)7-4-5-8(11)9(12)6-7/h4-6,11-12H,1-3H3
InChI 密鑰
XESZUVZBAMCAEJ-UHFFFAOYSA-N
尋找類似的產品? 前往 產品比較指南
一般說明
4-tert-Butylcatechol undergoes electrochemical trimerization via anodic oxidation and mechanism has been studied by cyclic voltammetry and controlled-potential coulometry. It inhibits the activity of tyrosinase at concentrations higher than 1×10−3M. It undergoes oxidation with laccase to yield quinones which on reaction with dienes and oxidation afford naphthoquinones.
應用
4-tert-Butylcatechol was used in the synthesis of tungsten oxide nanoparticles by nonaqueous sol-gel process.
訊號詞
Danger
危險分類
Acute Tox. 4 Dermal - Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1 - Skin Corr. 1B - Skin Sens. 1
儲存類別代碼
8A - Combustible corrosive hazardous materials
水污染物質分類(WGK)
WGK 3
閃點(°F)
235.4 °F - closed cup
閃點(°C)
113 °C - closed cup
個人防護裝備
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Y Usami et al.
Journal of toxicology and environmental health, 6(3), 559-567 (1980-05-01)
4-tert-Butylcatechol (TBC) is an antioxidant widely used in industry and a potent depigmenting agent to the skin of the workers. In this study, tyrosinase was extracted from tissue-cultured human melanoma cells and purified by polyacrylamide gel electrophoresis. T1 and T2
Ligand and solvent effects in the nonaqueous synthesis of highly ordered anisotropic tungsten oxide nanostructures.
Polleux J, et al.
Journal of Materials Chemistry, 16(40), 3969-3975 (2006)
Tetrahedron Letters, 48, 2983-2983 (2007)
Mechanistic study of electrochemical oxidation of 4-tert-butylcatechol: A facile electrochemical method for the synthesis of new trimer of 4-tert-butylcatechol.
Nematollahi D, et al.
Electrochimica Acta, 49(15), 2495-2502 (2004)
J N Rodriguez-López et al.
Analytical biochemistry, 202(2), 356-360 (1992-05-01)
A procedure for calibrating a Clark-type oxygen electrode is described. This method is based on the oxidation of 4-tert-butylcatechol (TBC) by O2 catalyzed by tyrosinase, to yield 4-tert-butyl-o-benzoquinone (TBCQ). This reaction consumes known amounts of oxygen in accordance with the
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務