T30805
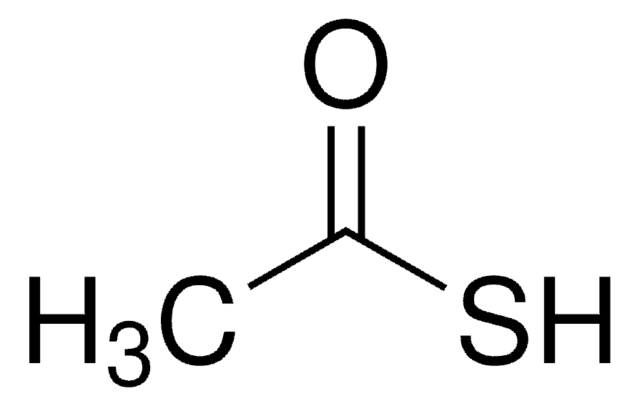
Thioacetic acid
96%
Synonym(s):
TAA, TMA, Thiacetic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CH3COSH
CAS Number:
Molecular Weight:
76.12
Beilstein:
1733298
EC Number:
MDL number:
UNSPSC Code:
12352106
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
96%
refractive index
n20/D 1.465 (lit.)
bp
88-91.5 °C (lit.)
density
1.065 g/mL at 25 °C (lit.)
storage temp.
2-8°C
SMILES string
CC(S)=O
InChI
1S/C2H4OS/c1-2(3)4/h1H3,(H,3,4)
InChI key
DUYAAUVXQSMXQP-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Thioacetic acid (TAA) can undergo:
- Enantioselective addition to nitroalkenes to form chiral 1,2-aminothiol derivatives in the presence of a novel sulfinyl urea organocatalyst. This method has been successfully employed in the synthesis of antifungal drug, sulconazole.
- Asymmetric Michael addition reaction with chalcones in the presence of a bifunctional amine thiourea catalyst to form synthetically useful thioesters.
- Asymmetric 1,6-conjugate addition with para-quinone methides in the presence of a chiral phosphoric acid catalyst to form chiral sulfur-containing diphenylmethane-type compounds.
- Conjugate addition to methacrylamides with chiral trans-2,5-disubstituted pyrrolidine auxiliaries to form chiral β-mercaptocarboxylic acid derivatives.
Thioacetic acid is a reagent for introduction of the thiol group into organic molecules.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Oral - Acute Tox. 4 Inhalation - Eye Dam. 1 - Flam. Liq. 2 - Skin Sens. 1
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
64.4 °F - closed cup
Flash Point(C)
18 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Phosphoric Acid Catalyzed Asymmetric 1, 6?Conjugate Addition of Thioacetic Acid to para?Quinone Methides.
Dong N, et al.
Angewandte Chemie (International Edition in English), 55(4), 1460-1464 (2016)
Organocatalytic enantioselective Michael addition of thioacetic acid to enones.
Li Hao, et al.
Tetrahedron Letters, 47(18), 3145-3148 (2006)
Asymmetric induction in the conjugate addition of thioacetic acid to methacrylamides with chiral auxiliaries.
Kim B H, et al.
Tetrahedron Asymmetry, 16(6), 1215-1220 (2005)
Christina Wedemeyer-Exl et al.
Organic & biomolecular chemistry, 5(13), 2119-2128 (2007-06-22)
The thiol-dependent methylation of heptamethyl cob(II)yrinate 8r with methyl iodide and methyl tosylate was explored under a variety of conditions. The interaction of the heptamethyl cob(II)yrinate with a variety of thiols was monitored prior to the addition of the methylating
Jianhua Ren
The journal of physical chemistry. A, 110(50), 13405-13411 (2006-12-15)
We studied the effects of polar groups on the gas-phase acidities of carboxylic acids experimentally and computationally. In this connection, the gas-phase acidities (DeltaH(acid), the enthalpy of deprotonation, and DeltaG(acid), the deprotonation free energy) of borane-complexed methylaminoacetic acid ((CH(3))2N(BH(3))CH(2)CO(2)H) and
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service