790796
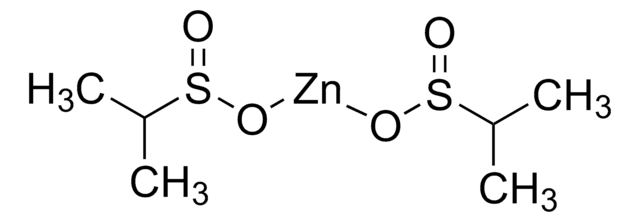
Zinc benzylsulfinate
Synonym(s):
Zinc benzylsulfinate, Baran BNS Reagent
About This Item
Recommended Products
form
solid
reaction suitability
reaction type: Alkylations
reaction type: C-C Bond Formation
reagent type: catalyst
reaction type: C-H Activation
reagent type: diversification reagent
mp
56 °C (Decomposition)
storage temp.
2-8°C
SMILES string
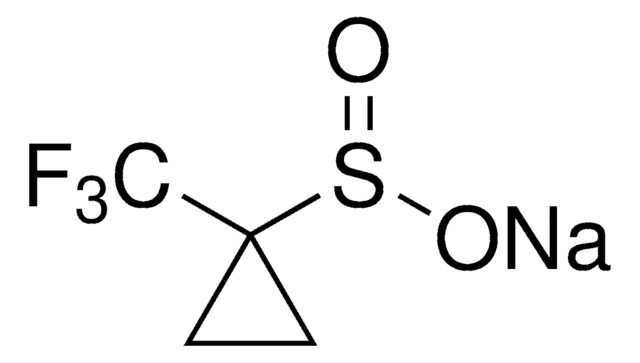
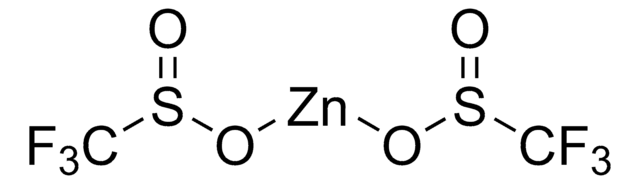
O=S(O[Zn]OS(CC1=CC=CC=C1)=O)CC2=CC=CC=C2
InChI
1S/2C7H8O2S.Zn/c2*8-10(9)6-7-4-2-1-3-5-7;/h2*1-5H,6H2,(H,8,9);/q;;+2/p-2
InChI key
AHSIXHPQMGLVIG-UHFFFAOYSA-L
Application
Practical and Innate Carbon-Hydrogen Functionalization of Heterocycles
Learn More at the Professor and Product Portal of Professor Phil S. Baran.
Other Notes
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Related Content
The synthesis of heteroaromatic and aromatic compounds is at the heart of the chemical industry. The ever-growing demand for new chemical entities, coupled with dwindling resources and time constraints allotted to any given research project, a rapid way to diversify (hetero)aromatic scaffolds is needed.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service