All Photos(1)
About This Item
Empirical Formula (Hill Notation):
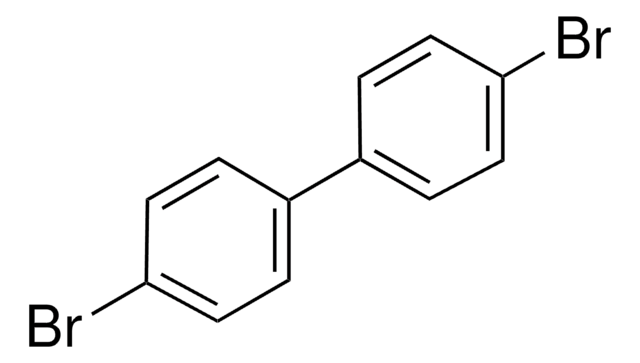
C24H15Br3
CAS Number:
Molecular Weight:
543.09
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
Assay
97%
form
solid
mp
261-265 °C
SMILES string
Brc1ccc(cc1)-c2cc(cc(c2)-c3ccc(Br)cc3)-c4ccc(Br)cc4
InChI
1S/C24H15Br3/c25-22-7-1-16(2-8-22)19-13-20(17-3-9-23(26)10-4-17)15-21(14-19)18-5-11-24(27)12-6-18/h1-15H
InChI key
HJQRITCAXSBOPC-UHFFFAOYSA-N
General description
1,3,5-Tris(4-bromophenyl)benzene (TBB) is a halogenated aromatic monomer that can be used in the formation of covalent aromatic frameworks(COF).
Application
TBB can be used to synthesize porous aromatic frameworks for the development of adsorption membranes to treat organic pollutants. It can also be used in the fabrication of pyridine based high efficiency organic light emitting diodes(OLEDs).
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 4 - Eye Dam. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Surface mediated synthesis of 2D covalent organic frameworks: 1, 3, 5-tris (4-bromophenyl) benzene on graphite (001), Cu (111), and Ag (110)
Gutzler R, et al.
Chemical Communications (Cambridge, England), 4456-4458 (2009)
Star-shaped oligothiophenes for solution-processible organic electronics: flexible aliphatic spacers approach
Ponomarenko SA, et al.
Chemistry of Materials, 18(17), 4101-4108 (2006)
Pyridine-containing triphenylbenzene derivatives with high electron mobility for highly efficient phosphorescent OLEDs
Su, Shi-Jian; Chiba, Takayuki; et al.
Advanced Materials, 20(11), 2125-2130 (2008)
Synthesis of a porous aromatic framework for adsorbing organic pollutants application
Ren H, et al.
Journal of Materials Chemistry, 21(28), 10348-10353 (2011)
Ruiyan Sun et al.
ChemSusChem, 12(14), 3278-3285 (2019-04-30)
Methyl formate was produced in one pot through the hydrogenation of CO2 to formic acid/formate followed by an esterification step. The route offers the possibility to integrate renewable energy into the fossil-based chemical value chain. In this work, a phosphine-polymer-anchored
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service