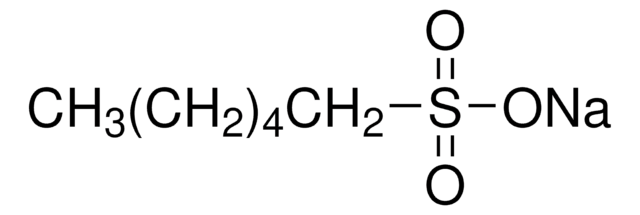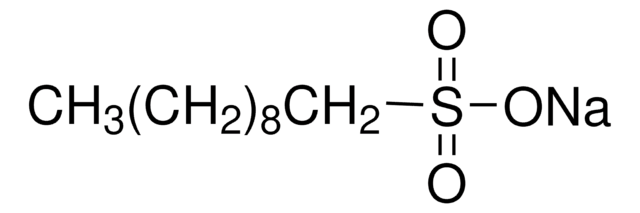H9026
Sodium hexanesulfonate
BioXtra
Synonym(s):
1-Hexanesulfonic acid sodium salt
About This Item
Recommended Products
description
anionic
Quality Level
product line
BioXtra
mol wt
188.22 g/mol
technique(s)
HPLC: suitable
impurities
<0.001% Phosphorus (P)
<0.1% Insoluble matter
solubility
H2O: 0.1 M at 20 °C, clear, colorless
cation traces
Al: <0.0005%
Ca: <0.0005%
Cu: <0.0005%
Fe: <0.0005%
K: <0.005%
Mg: <0.0005%
NH4+: <0.05%
Pb: <0.001%
Zn: <0.0005%
SMILES string
[Na+].CCCCCCS([O-])(=O)=O
InChI
1S/C6H14O3S.Na/c1-2-3-4-5-6-10(7,8)9;/h2-6H2,1H3,(H,7,8,9);/q;+1/p-1
InChI key
QWSZRRAAFHGKCH-UHFFFAOYSA-M
Looking for similar products? Visit Product Comparison Guide
Application
<li><strong>Ion pair assisted micro matrix solid phase dispersion extraction of alkaloids from medical plant</strong>: Sodium hexanesulfonate was used as an ion-pairing reagent in the extraction of alkaloids for pharmacological studies, demonstrating its efficacy in enhancing the selectivity and sensitivity of HPLC methods in the analysis of complex plant matrices (Dong et al., 2020).</li>
<li><strong>Pharmacokinetic Properties of Arsenic Species after Intravenous and Intragastrical Administration of Arsenic Trioxide Solution in Cynomolgus Macaques Using HPLC-ICP-MS</strong>: This study highlights the role of Sodium hexanesulfonate in improving the chromatographic profiles of arsenic species, which is crucial for accurate pharmacokinetic assessment in pharmaceutical research (Shi et al., 2019).</li>
<li><strong>Organic solvent-free reversed-phase ion-pairing liquid chromatography coupled to atomic fluorescence spectrometry for organoarsenic species determination in several matrices</strong>: Sodium hexanesulfonate is utilized as a solvent-free ion-pairing agent, enabling greener analytical processes without compromising the performance of liquid chromatography systems (Monasterio et al., 2011).</li>
</ul>
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service