8.52078
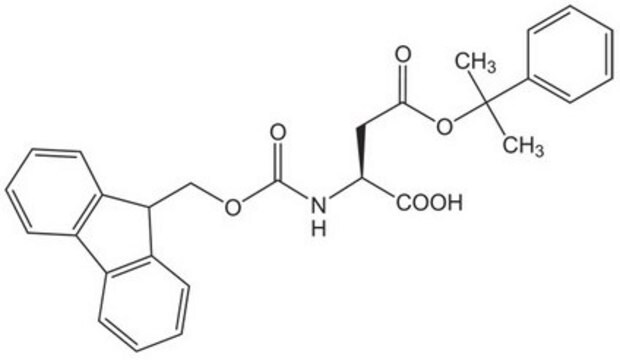
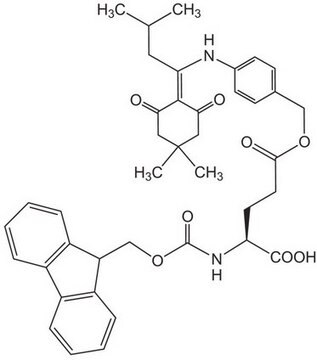
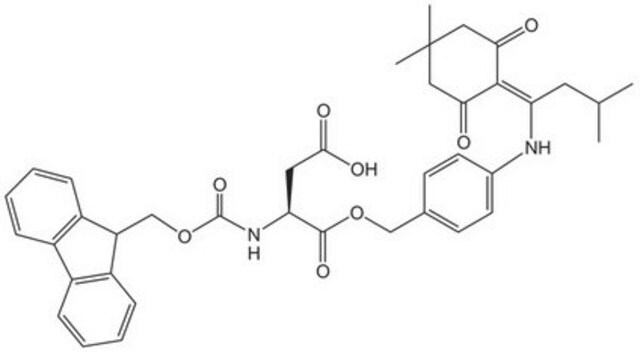
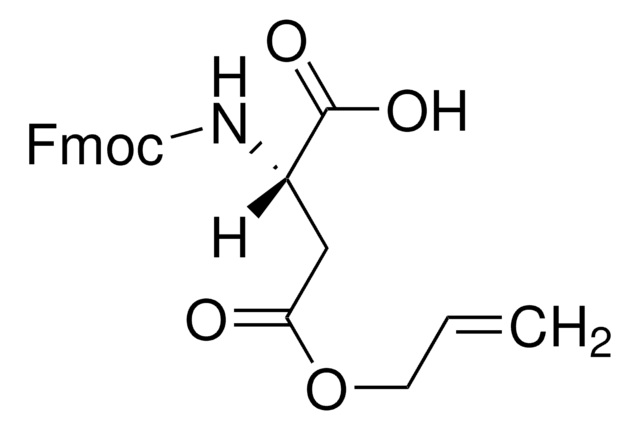
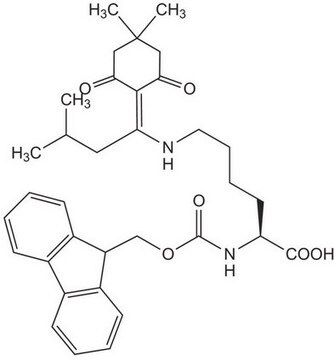
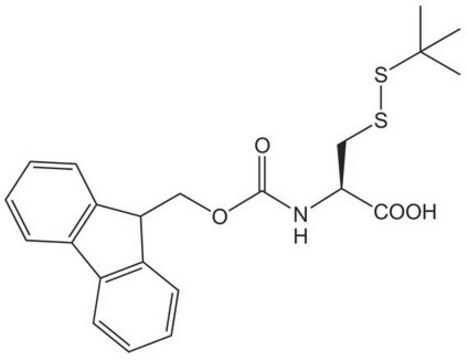
Fmoc-Asp(ODmab)-OH
≥98% (TLC), for peptide synthesis, Novabiochem®
Synonym(s):
Fmoc-Asp(ODmab)-OH, N-α-Fmoc-L-aspartic acid β-4-{N-[1-(4,4-dimethyl-2,6-dioxocyclohexylidene)-3-methylbutyl]-amino} benzyl ester
About This Item
Recommended Products
product name
Fmoc-Asp(ODmab)-OH, Novabiochem®
Quality Level
product line
Novabiochem®
Assay
≥96.0% (HPLC)
≥98% (TLC)
form
powder
reaction suitability
reaction type: Fmoc solid-phase peptide synthesis
manufacturer/tradename
Novabiochem®
application(s)
peptide synthesis
functional group
carboxylic acid
storage temp.
15-25°C
InChI
1S/C39H42N2O8/c1-23(2)17-31(36-33(42)19-39(3,4)20-34(36)43)40-25-15-13-24(14-16-25)21-48-35(44)18-32(37(45)46)41-38(47)49-22-30-28-11-7-5-9-26(28)27-10-6-8-12-29(27)30/h5-16,23,30,32,40H,17-22H2,1-4H3,(H,41,47)(H,45,46)/t32-/m0/s1
InChI key
FLMMHDDOOIGEPG-YTTGMZPUSA-N
General description
Associated Protocols and Technical Articles
Cleavage and Deprotection Protocols for Fmoc SPPS
Literature references
[1] W. C. Chan, et al. (1995) J. Chem. Soc., Chem. Commun., 2209.
Linkage
Analysis Note
Appearance of substance (visual): powder
Identity (IR): passes test
Enantiomeric purity: ≥ 99.5 % (a/a)
Purity (TLC(157B)): ≥ 98 %
Purity (TLC(CMA2)): ≥ 98 %
Assay (HPLC, area%): ≥ 96.0 % (a/a)
Solubility (1 mmole in 2 ml DMF): clearly soluble
Water (K. F.): ≤ 1.0 %
To see the solvent systems used for TLC of Novabiochem® products please click here.
Legal Information
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Novabiochem® product range has one of the largest collections of orthogonally and quasi-orthogonally protected tri-functional amino acids. These derivatives are useful tools for the synthesis of cyclic and branched peptides and peptides carrying side-chain modifications.
Protocols
Novabiochem® product range has one of the largest collections of orthogonally and quasi-orthogonally protected tri-functional amino acids. These derivatives are useful tools for the synthesis of cyclic and branched peptides and peptides carrying side-chain modifications
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service