8.52017
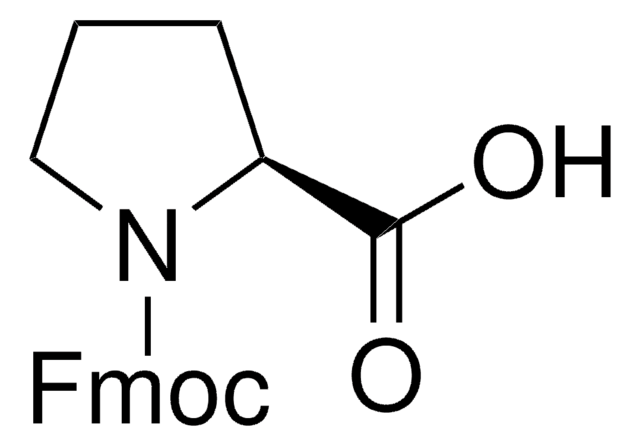
Fmoc-Pro-OH
Novabiochem®
Synonym(s):
Fmoc-Pro-OH, N-α-Fmoc-L-proline
About This Item
Recommended Products
Quality Level
product line
Novabiochem®
Assay
≥94.0% (acidimetric)
≥98% (TLC)
≥99.0% (HPLC)
form
powder
reaction suitability
reaction type: Fmoc solid-phase peptide synthesis
manufacturer/tradename
Novabiochem®
mp
112-115 °C
application(s)
peptide synthesis
functional group
Fmoc
storage temp.
2-30°C
InChI
1S/C20H19NO4/c22-19(23)18-10-5-11-21(18)20(24)25-12-17-15-8-3-1-6-13(15)14-7-2-4-9-16(14)17/h1-4,6-9,17-18H,5,10-12H2,(H,22,23)
InChI key
ZPGDWQNBZYOZTI-UHFFFAOYSA-N
General description
Standard building block for introduction of proline amino-acid residues by Fmoc SPPS
Associated Protocols and Technical Articles
Fmoc-amino acids for Peptide Production
Cleavage and Deprotection Protocols for Fmoc SPPS
Application
- Biphasic electrochemical peptide synthesis: This study highlights the application of Fmoc-Pro-OH in electrochemical peptide synthesis, where slight excesses of reagents and electricity were required, possibly due to steric hindrance (S Nagahara, Y Okada, Y Kitano, K Chiba - Chemical Science, 2021).
- Elucidation of the Mechanism of Endo-XaaC-terminal Peptide Impurity Formation in SPPS: Fmoc-Pro-OH′s role was analyzed in a study investigating impurity formation during solid-phase peptide synthesis, highlighting its distinctively low tendency to form impurities (Y Yang, L Hansen, A Baldi - Organic Process Research & Development, 2021).
- Polymer–peptide delivery platforms: This research utilized Fmoc-Pro-OH in the development of polymer-based DNA delivery systems, emphasizing the effect of oligopeptide orientation on delivery efficacy (SS Parelkar, R Letteri, D Chan-Seng - Biomacromolecules, 2014).
- Structure–Activity Relationship Study of N-Hydroxyphtalimide Derivatives: The study explored the use of Fmoc-Pro-OH in the synthesis of N-Hydroxyphtalimide derivatives for detecting amines during peptide synthesis (K Takamatsu, R Suzuki, A Matsunaga - The Journal of Organic Chemistry, 2023).
Linkage
Analysis Note
Appearance of substance (visual): powder
Colour index (0,5 M in DMF): ≤ 150 Hazen
Identity (IR): passes test
Enantiomeric purity: ≥ 99.8 % (a/a)
Purity (HPLC): ≥ 99.0 % (a/a)
Fmoc-ß-Ala-OH (HPLC): ≤ 0.1 % (a/a)
Fmoc-β-Ala-Pro-OH (HPLC): ≤ 0.1 % (a/a)
Fmoc-Pro-Pro-OH (HPLC): ≤ 0.1 % (a/a)
Assay free amino acid (GC): ≤ 0.2 %
Purity (TLC(011A)): ≥ 98 %
Solubility (25 mmole in 50 ml DMF): clearly soluble
Assay (acidimetric): ≥ 94.0 %
Water (K. F.): ≤ 6.0 %
Ethyl acetate (HS-GC): ≤ 0.5 %
Acetate (IC): ≤ 0.02 %
To see the solvent systems used for TLC of Novabiochem® products please click here.
Legal Information
Not finding the right product?
Try our Product Selector Tool.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Related Content
Purer Fmocs Means Purer Peptides
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service