515477
4,4′-Di-tert-butyl-2,2′-dipyridyl
98%
Synonym(s):
4,4′-Bis(tert-butyl)-2,2′-bipyridine, BBBPY
About This Item
Recommended Products
Assay
98%
mp
159-161 °C (lit.)
SMILES string
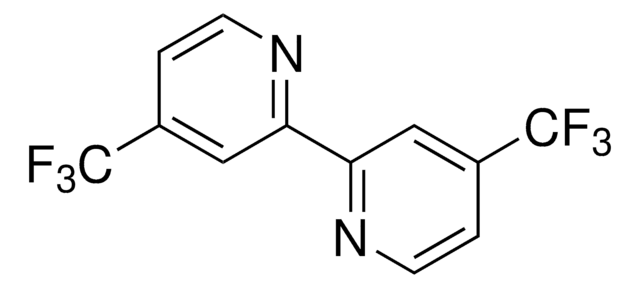
CC(C)(C)c1ccnc(c1)-c2cc(ccn2)C(C)(C)C
InChI
1S/C18H24N2/c1-17(2,3)13-7-9-19-15(11-13)16-12-14(8-10-20-16)18(4,5)6/h7-12H,1-6H3
InChI key
TXNLQUKVUJITMX-UHFFFAOYSA-N
Related Categories
General description
Application
- In the synthesis of oxidovanadium(IV) complex [VOCl2(dbbpy)(H2O)], which is used as a catalyst for the epoxidation of cyclooctene in the presence of tert-butyl hydroperoxide.
- In iron-catalyzed ortho-allylation of 1-arylpyrazoles via C-H activation.
- In iridium-catalyzed borylation of (hetero)arenes.
- In iron-catalyzed arylation of heterocycles in the presence of metallic Mg.
- In nickel-catalyzed decarboxylation of block copolymers.
related product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Oral
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Tools and techniques for performing atom transfer radical polymerization (ATRP) with benefits and limitations.
Arylboronic acids and esters are invaluable tools for the chemical community. These powerful reagents are used for a variety of transformations, most notably the Suzuki-Miyaura cross-coupling reaction.
We presents an article about a micro review of reversible addition/fragmentation chain transfer (RAFT) polymerization. RAFT (Reversible Addition/Fragmentation Chain Transfer) polymerization is a reversible deactivation radical polymerization (RDRP) and one of the more versatile methods for providing living characteristics to radical polymerization.
Tools for Performing ATRP
Protocols
We presents an article featuring procedures that describe polymerization of methyl methacrylate and vinyl acetate homopolymers and a block copolymer as performed by researchers at CSIRO.
We present an article about RAFT, or Reversible Addition/Fragmentation Chain Transfer, which is a form of living radical polymerization.
An article about the typical procedures for polymerizing via ATRP, which demonstrates that in the following two procedures describe two ATRP polymerization reactions as performed by Prof. Dave Hadddleton′s research group at the University of Warwick.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service

![(Ir[dF(CF3)ppy]2(dtbpy))PF6](/deepweb/assets/sigmaaldrich/product/structures/982/913/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09/640/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09.png)
![[4,4′-Bis(1,1-dimethylethyl)-2,2′-bipyridine] nickel (II) dichloride](/deepweb/assets/sigmaaldrich/product/structures/471/091/6faa29b1-bf8a-4d87-90b2-4cc55e082620/640/6faa29b1-bf8a-4d87-90b2-4cc55e082620.png)
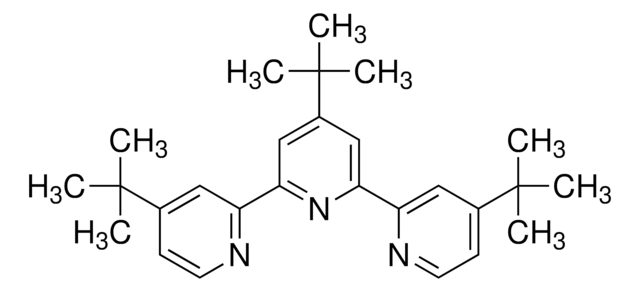

![[Ir(dtbbpy)(ppy)2]PF6](/deepweb/assets/sigmaaldrich/product/structures/158/329/2544d673-d267-4aa1-8f46-2652aad4bfa0/640/2544d673-d267-4aa1-8f46-2652aad4bfa0.png)

![[Ir(dF(Me)ppy)2(dtbbpy)]PF6](/deepweb/assets/sigmaaldrich/product/structures/150/099/7c2dfa31-39f4-4cca-aee5-86d4a89fea78/640/7c2dfa31-39f4-4cca-aee5-86d4a89fea78.png)