451657
Copper(II) sulfate
anhydrous, powder, ≥99.99% trace metals basis
Synonym(s):
Cupric sulfate
About This Item
Recommended Products
grade
anhydrous
Quality Level
vapor pressure
7.3 mmHg ( 25 °C)
Assay
≥99.99% trace metals basis
form
powder
impurities
≤100.0 ppm Trace Metal Analysis
mp
200 °C (dec.) (lit.)
density
3.603 g/mL at 25 °C (lit.)
application(s)
battery manufacturing
SMILES string
[Cu++].[O-]S([O-])(=O)=O
InChI
1S/Cu.H2O4S/c;1-5(2,3)4/h;(H2,1,2,3,4)/q+2;/p-2
InChI key
ARUVKPQLZAKDPS-UHFFFAOYSA-L
Looking for similar products? Visit Product Comparison Guide
General description
Application
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
In many technologies, performance requirements drive device dimensions below the scale of electron mean free paths (λe). This trend has increased scientific interest and technological importance of electrical resistivities at the nanoscale.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
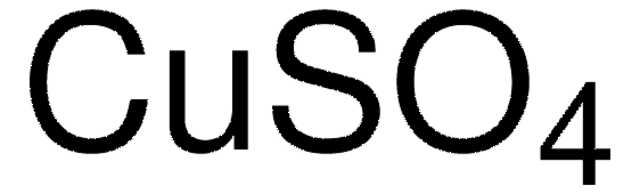







![Tris[(1-benzyl-1H-1,2,3-triazol-4-yl)methyl]amine 97%](/deepweb/assets/sigmaaldrich/product/structures/179/695/86a721c8-2a4c-4e4f-bc36-6276ce7a941f/640/86a721c8-2a4c-4e4f-bc36-6276ce7a941f.png)


