173282
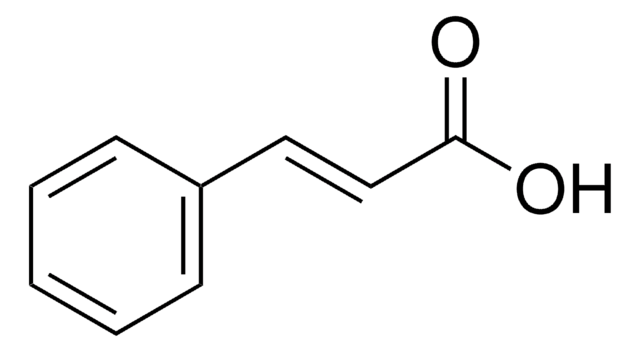
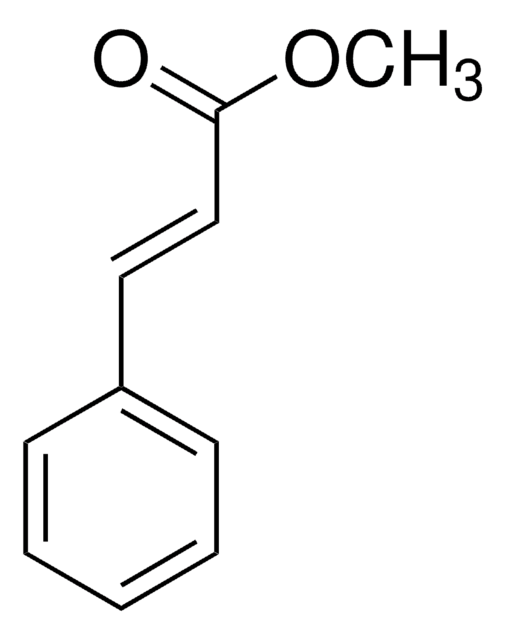
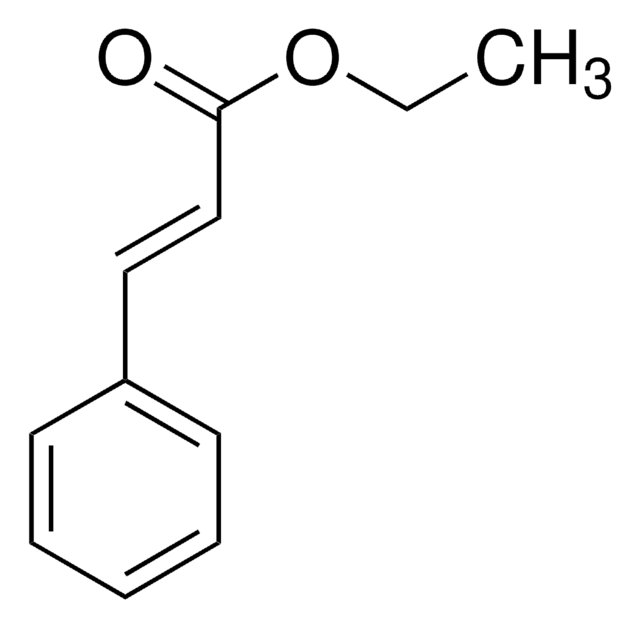
Methyl trans-cinnamate
99%
Synonym(s):
trans-Cinnamic acid methyl ester
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
C6H5CH=CHCO2CH3
CAS Number:
Molecular Weight:
162.19
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
form
solid
bp
260-262 °C (lit.)
mp
34-38 °C (lit.)
SMILES string
COC(=O)\C=C\c1ccccc1
InChI
1S/C10H10O2/c1-12-10(11)8-7-9-5-3-2-4-6-9/h2-8H,1H3/b8-7+
InChI key
CCRCUPLGCSFEDV-BQYQJAHWSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Methyl trans-cinnamate has antimicrobial ability.
Application
Methyl trans-cinnamate was used to inhibit monophenolase and diphenolase activity of mushroom tyrosinase.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Marta Jiménez-Salcedo et al.
Journal of chromatography. A, 1487, 14-21 (2017-01-29)
Specific blends of essential oils (BEOs) are promising substitutes for antibiotics to promote livestock performance and to reduce the incidence of intestinal disorders. Microencapsulation of BEOs has shown to improve their stability, bioavailability and to control their release rate once
Vigilio Ballabeni et al.
Fitoterapia, 81(4), 289-295 (2009-10-15)
Here we investigated the anti-inflammatory properties of Ocotea quixos essential oil and of its main components, trans-cinnamaldehyde and methyl cinnamate, in in vitro and in vivo models. Ocotea essential oil and trans-cinnamaldehyde but not methyl cinnamate significantly reduced LPS-induced NO
Kathrin Fink et al.
Journal of agricultural and food chemistry, 52(10), 3065-3068 (2004-05-13)
For the authenticity assessment of (E)-methyl cinnamate from different origins, combustion/pyrolysis-isotope ratio mass spectrometry (C/P-IRMS) was used by an elemental analyzer (EA) and on-line capillary gas chromatography coupling (HRGC-C/P-IRMS). For that reason, (E)-methyl cinnamate self-prepared from synthetic, natural, and semisynthetic
Valtcho D Zheljazkov et al.
Journal of agricultural and food chemistry, 56(1), 241-245 (2007-12-13)
A field experiment was conducted to assess yield, oil content, and composition of 38 genotypes of sweet basil ( Ocimum basilicum L.). Overall, biomass yields were high and comparable to those reported in the literature. However, basil genotypes differed significantly
Takuo Sawahata et al.
Mycorrhiza, 18(2), 111-114 (2007-12-11)
Two major volatiles produced by the mycelia and fruiting bodies of Tricholoma matsutake (1-octen-3-ol and methyl cinnamate) repel a mycophagous collembolan, Proisotoma minuta. Aggregation of the collembolans on their diet was significantly inhibited by exposure to 1 ppm methyl cinnamate
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service