392251
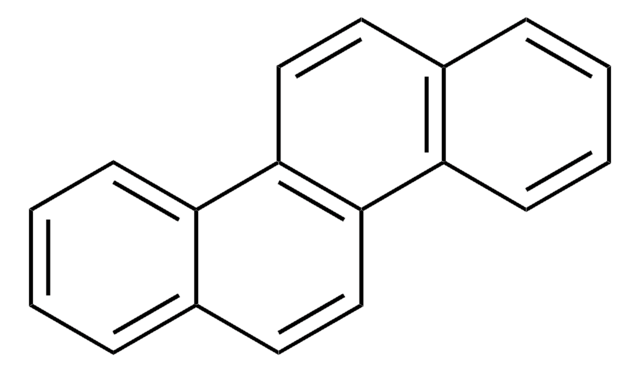
Benzo[k]fluoranthene
for fluorescence, ≥99%
Synonym(s):
11,12-Benzofluoranthene, 2,3,1′,8′-Binaphthylene, 8,9-Benzfluoranthene
About This Item
Recommended Products
grade
for fluorescence
Quality Level
Assay
≥99%
mp
215-217 °C (lit.)
solubility
95% ethanol: <1 mg/mL at 20 °C
DMSO: <1 mg/mL at 20 °C
H2O: <1 mg/mL at 20 °C
acetone: 1-10 mg/mL at 20 °C
methanol: <1 mg/mL at 20 °C
toluene: 5-10 mg/mL at 20 °C
SMILES string
c1ccc2cc-3c(cc2c1)-c4cccc5cccc-3c45
InChI
1S/C20H12/c1-2-6-15-12-19-17-10-4-8-13-7-3-9-16(20(13)17)18(19)11-14(15)5-1/h1-12H
InChI key
HAXBIWFMXWRORI-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- Enhanced degradation of carcinogenic PAHs benzo (a) pyrene and benzo (k) fluoranthene by a microbial consortia: Bioremediation of high molecular weight PAHs with a combination of microorganisms (S Guntupalli, V Thunuguntla, 2016).
- Biotransformation of the high‐molecular weight polycyclic aromatic hydrocarbon (PAH) benzofluoranthene by Sphingobium sp. strain KK22 and identification of metabolites: Biotransformation and identification of products from benzo[k]fluoranthene (AH Maeda, S Nishi, Y Hatada, Y Ozeki, 2014).
- Investigation of the electrochemical properties of benzofluorenthene using a glassy carbon electrode and development of a square-wave voltammetric method for detection: Electrochemical behavior of benzo[k]fluorenthene and development of detection method (A Altun, Y Yardim, A Levent, 2023).
Packaging
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 1B
Storage Class Code
6.1C - Combustible, acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![Benzo[b]fluoranthene 98%](/deepweb/assets/sigmaaldrich/product/structures/175/744/6fa5fca2-b6ec-47b6-ab7a-fe895843f226/640/6fa5fca2-b6ec-47b6-ab7a-fe895843f226.png)
![Benzo[ghi]perylene 98%](/deepweb/assets/sigmaaldrich/product/structures/154/740/c50ff1be-dfb4-4159-a98c-9cecf9206ad3/640/c50ff1be-dfb4-4159-a98c-9cecf9206ad3.png)
![Indeno[1,2,3-cd]pyrene analytical standard](/deepweb/assets/sigmaaldrich/product/structures/231/153/b0b230c2-efa0-4f43-a261-66b931ead3d2/640/b0b230c2-efa0-4f43-a261-66b931ead3d2.png)
![Dibenz[a,h]anthracene certified reference material, TraceCERT®, Manufactured by: Sigma-Aldrich Production GmbH, Switzerland](/deepweb/assets/sigmaaldrich/product/structures/358/871/0a80ecfc-d123-44ca-90a6-22248b43aba9/640/0a80ecfc-d123-44ca-90a6-22248b43aba9.png)
![Benzo[a]pyrene ≥96% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/253/820/be96d879-1811-46c0-8f11-612019691c2d/640/be96d879-1811-46c0-8f11-612019691c2d.png)

![Benzo[k]fluoranthene certified reference material, TraceCERT®, Manufactured by: Sigma-Aldrich Production GmbH, Switzerland](/deepweb/assets/sigmaaldrich/product/structures/277/320/3e615f9f-3887-40f6-b176-bc1eb9b4832c/640/3e615f9f-3887-40f6-b176-bc1eb9b4832c.png)