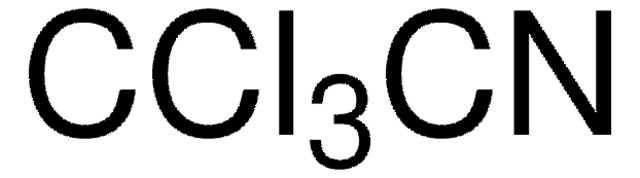All Photos(2)
About This Item
Linear Formula:
H2NNH2 · CH3CO2H
CAS Number:
Molecular Weight:
92.10
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
crystals
mp
100-102 °C (lit.)
functional group
carboxylic acid
hydrazine
SMILES string
NN.CC(O)=O
InChI
1S/C2H4O2.H4N2/c1-2(3)4;1-2/h1H3,(H,3,4);1-2H2
InChI key
YFHNDHXQDJQEEE-UHFFFAOYSA-N
Related Categories
General description
Hydrazine acetate is an acetate salt of hydrazine. It is used as a reagent for the cleavage of glycosidic esters and anomeric denitration of carbohydrates. In organic synthesis, it is also used as a building block to prepare various organic derivatives.
Application
Hydrazine acetate has been used in preparation of:
- 2,3,5-tri-O-acetyl-α-L-arabinofuranosyl trichloroacetimidate
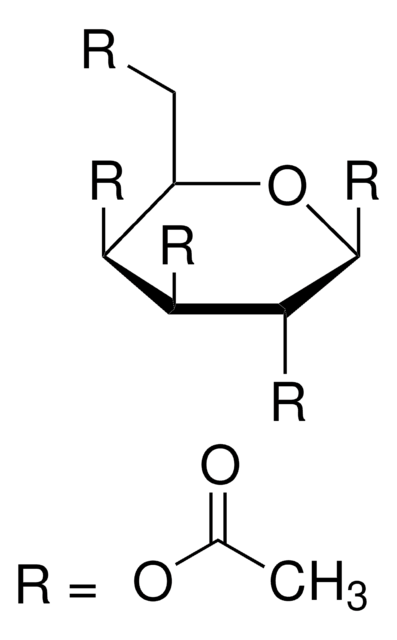
- disaccharide 4-methoxyphenyl glycoside
related product
Product No.
Description
Pricing
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 1B - Skin Sens. 1
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
S Deng et al.
Carbohydrate research, 317(1-4), 53-62 (1999-08-31)
Dioscin, polyphyllin D, and balanitin 7, which belong to a group of structurally similar diosgenyl saponins with promising bioactivities, were synthesized by stepwise glycosylation.
T M Slaghek et al.
Carbohydrate research, 255, 61-85 (1994-03-04)
The synthesis is reported of 4-methoxyphenyl O-(beta-D-glucopyranosyluronic acid)-(1-->3)-2-acetamido-2-deoxy-beta-D-glucopyranoside (1), 4-methoxyphenyl O-(2-acetamido-2-deoxy-beta-D-glucopyranosyl)-(1-->4)-O-(beta-D- glucopyranosyluronic acid)-(1-->3)-2-acetamido-2-deoxy-beta-D-glucopyranoside (5), and 4-methoxyphenyl O-(beta-D-glucopyranosyluronic acid)-(1-->3)-O-(2-acetamido-2-deoxy-beta-D-glucopyranosyl)-(1-->4)-O-(b eta-D- glucopyranosyluronic acid)-(1-->3)-2-acetamido-2-deoxy-beta-D-glucopyranoside (10), which are structural elements of the extracellular polysaccharide hyaluronic acid. 6-O-Levulinoyl-2,3,4-tri-O-p-toluoyl-alpha-D-glucopyranosyl trichloroacetimidate (3) was condensed with 4-methoxyphenyl 2-deoxy-4,6-O-isopropylidene-2-phthalimido-beta-D-glucopyranoside
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service

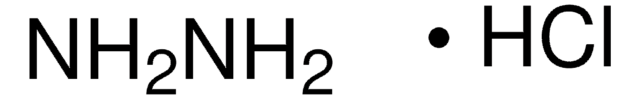


![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)