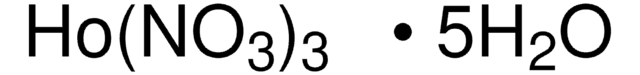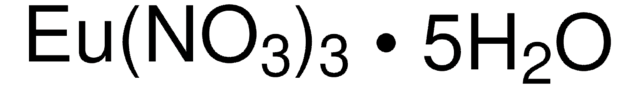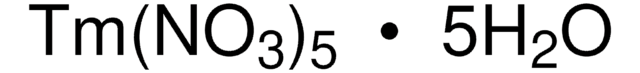205133
Praseodymium(III) nitrate hexahydrate
99.9% trace metals basis
Synonym(s):
Praseodymium trinitrate hexahydrate
About This Item
Assay
99.9% trace metals basis
form
crystalline
reaction suitability
reagent type: catalyst
core: praseodymium
impurities
≤2000 ppm Trace Metal Analysis
SMILES string
O.O.O.O.O.O.[Pr+3].[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O
InChI
1S/3NO3.6H2O.Pr/c3*2-1(3)4;;;;;;;/h;;;6*1H2;/q3*-1;;;;;;;+3
InChI key
LXXCECZPOWZKLC-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- A dopant to fabricate dye-sensitized solar cells. The addition of rare earth enhances the power conversion efficiency of solar cells by narrowing the band gap of photoanode materials.
- A precursor to synthesize high entropy lanthanide oxysulfides ( wide band gap semiconductors).
- To synthesize functionalized UV-emitting nanocomposite for photodynamic cancer therapy.
- To fabricate Pr-doped MoO3 thinfilms for gas sensing applications.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1 - Eye Irrit. 2 - Ox. Sol. 3 - Skin Irrit. 2
Storage Class Code
5.1B - Oxidizing hazardous materials
WGK
WGK 2
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Innovation in dental restorative materials is driven by the need for biocompatible and natural-appearing restoration alternatives. Conventional dental materials like amalgam and composite resins have inherent disadvantages.
The rare earth elements impact nearly everyone in the world. All of the people living in advanced technological countries and almost all those living in third world countries utilize the rare earths in their everyday living—the car that one drives (gasoline is refined from oil using rare earth catalysts and catalytic converters reduce the polluting emissions from the automotive exhaust), watching the news on TV (the red and green colors in TV screens), the telephones and computers we use to communicate (the permanent magnets in speakers and disc drives), just to name a few examples.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service