133760
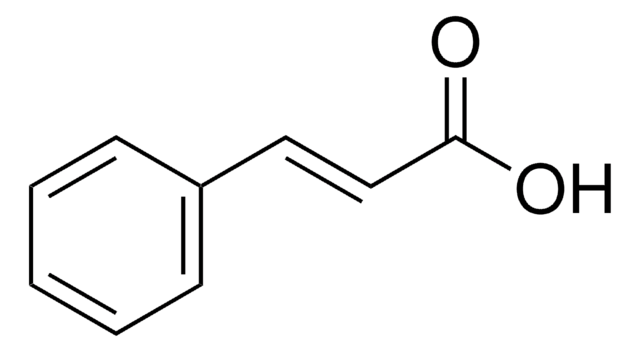
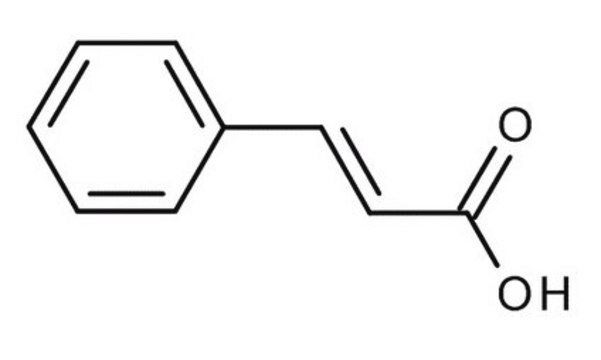
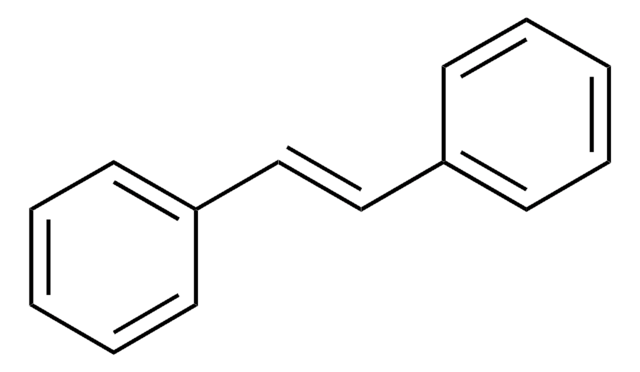
trans-Cinnamic acid
97%
Synonym(s):
trans-3-Phenylacrylic acid, Cinnamic acid
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
C6H5CH=CHCOOH
CAS Number:
Molecular Weight:
148.16
Beilstein:
1905952
EC Number:
MDL number:
UNSPSC Code:
12352100
eCl@ss:
39023931
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
solid
bp
300 °C (lit.)
mp
132-135 °C (lit.)
functional group
carboxylic acid
phenyl
SMILES string
OC(=O)\C=C\c1ccccc1
InChI
1S/C9H8O2/c10-9(11)7-6-8-4-2-1-3-5-8/h1-7H,(H,10,11)/b7-6+
InChI key
WBYWAXJHAXSJNI-VOTSOKGWSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
trans-Cinnamic acid was used to establish library of phenolic compounds by liquid chromatography/ultraviolet and mass spectrometry/mass spectrometry.
Biochem/physiol Actions
trans-cinnamic acid has inhibitory effect on phorbol-12-myristate-13-acetate-induced invasion of human lung adenocarcinoma A549 cells. It is a potential agent which can prevent lung tumor cells from metastasizing. It induces intracellular release of Ca2+ from the vacuole to the cytoplasm which triggers phytotoxicity in cucumber.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 1
Flash Point(F)
320.0 °F - closed cup
Flash Point(C)
160 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Jianping Sun et al.
Molecules (Basel, Switzerland), 12(3), 679-693 (2007-09-14)
Liquid chromatography/ultraviolet (LC/UV) and mass spectrometry/mass spectrometry (MS/MS) libraries containing 39 phenolic compounds were established by coupling a LC and an ion trap MS with an electrospray ionization (ESI) source, operated in negative ion mode. As a result, the deprotonated
Chiung-Man Tsai et al.
European journal of pharmaceutical sciences : official journal of the European Federation for Pharmaceutical Sciences, 48(3), 494-501 (2012-12-12)
Dietary polyphenols have been reported as an effective phytochemical for health protection and cinnamic acid (CA) is one of the polyphenols that has been demonstrated having chemopreventive potential. It was known that the early and distal metastasis might lead to
Jingquan Yu et al.
Journal of chemical ecology, 35(12), 1471-1477 (2010-01-12)
To obtain insight into interspecies interactions mediated by allelochemicals, the response of cucumber (Cucumis sativus L. cv Jinyan No.4) and figleaf gourd (Cucurbita ficifolia Bouché) seedlings to trans-cinnamic acid (CA) (1) was investigated. While trans-CA is an autotoxin in cucumber
Feng Yang et al.
Molecular pharmaceutics, 9(11), 3259-3265 (2012-09-27)
Owing to advantageous biochemical and pharmacological properties of human serum albumin (HSA), HSA-based drug carrier is playing an increasing role in the clinical setting. Since the IIA subdomain of HSA is a big hydrophobic cavity, we proposed that HSA delivers
Andrew M Lauer et al.
Organic letters, 14(19), 5138-5141 (2012-09-25)
A highly regioselective, Pd-catalyzed allylic fluorination of phosphorothioate esters is reported. This chemistry addresses several limitations of previously reported methods in which elimination and lack of reactivity were problematic. Preliminary mechanistic investigations reveal that these reactions are stereospecific and provide
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 133760-100G | 4061838729644 |
| 133760-2KG | 4061831828276 |
| 133760-500G | 4061838729651 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service