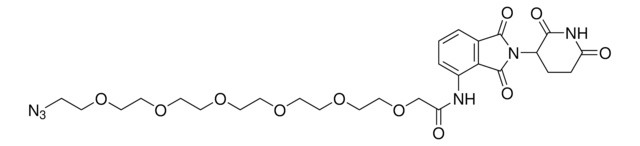
929336
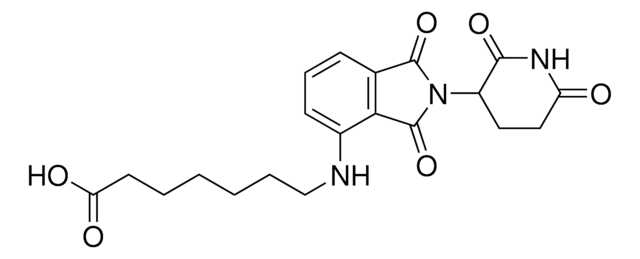
Pomalidomide-C5-phosphoramidite
Synonym(s):
2-Cyanoethyl (5-((2-(2,6-dioxopiperidin-3-yl)-1,3-dioxoisoindolin-4-yl)amino)pentyl) diisopropylphosphoramidite
About This Item
Recommended Products
ligand
pomalidomide
Quality Level
form
powder
storage temp.
2-8°C
SMILES string
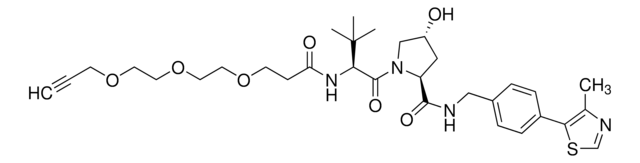
O=C1C2=C(C(NCCCCCOP(OCCC#N)N(C(C)C)C(C)C)=CC=C2)C(N1C3C(NC(CC3)=O)=O)=O
InChI
1S/C27H38N5O6P/c1-18(2)32(19(3)4)39(38-17-9-14-28)37-16-7-5-6-15-29-21-11-8-10-20-24(21)27(36)31(26(20)35)22-12-13-23(33)30-25(22)34/h8,10-11,18-19,22,29H,5-7,9,12-13,15-17H2,1-4H3,(H,30,33,34)
InChI key
LDWPFUQTEOHLBX-UHFFFAOYSA-N
Application
Technology Spotlight: Degrader Building Blocks for Targeted Protein Degradation
Protein Degrader Building Blocks
Other Notes
Destruction of DNA-Binding Proteins by Programmable Oligonucleotide PROTAC (O′PROTAC): Effective Targeting of LEF1 and ERG
Small-Molecule PROTACS: New Approaches to Protein Degradation
Targeted Protein Degradation: from Chemical Biology to Drug Discovery
Impact of linker length on the activity of PROTACs
Legal Information
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Repr. 1B
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documents section.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service