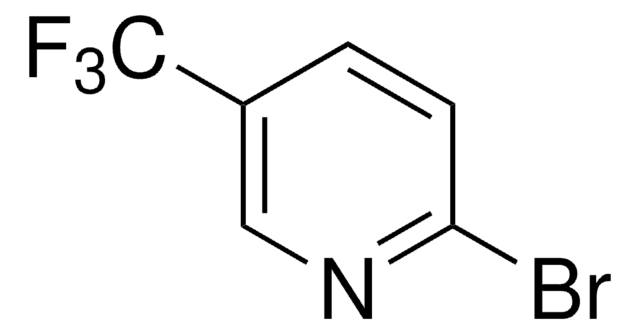
909335
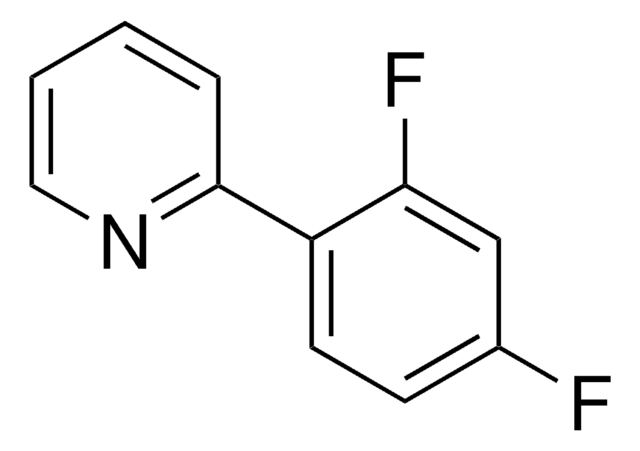
2-(2,4-Difluorophenyl)-5-(trifluoromethyl)pyridine
≥95%
Synonym(s):
dFCF3ppy
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C12H6F5N
CAS Number:
Molecular Weight:
259.17
MDL number:
UNSPSC Code:
12352101
NACRES:
NA.22
Recommended Products
Assay
≥95%
form
powder or crystals
reaction suitability
reaction type: Photocatalysis
reagent type: catalyst
mp
59-64 °C
InChI
1S/C12H6F5N/c13-8-2-3-9(10(14)5-8)11-4-1-7(6-18-11)12(15,16)17/h1-6H
InChI key
FMKQPMDFNYNYAG-UHFFFAOYSA-N
Application
2-(2,4-Difluorophenyl)-5-(trifluoromethyl)pyridine is a ligand used for the preparation of Ir(III) photocatalysts.
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Kazimer L Skubi et al.
Journal of the American Chemical Society, 139(47), 17186-17192 (2017-11-01)
Stereochemical control of electronically excited states is a long-standing challenge in photochemical synthesis, and few catalytic systems that produce high enantioselectivities in triplet-state photoreactions are known. We report herein an exceptionally effective chiral photocatalyst that recruits prochiral quinolones using a
Thomas Rossolini et al.
Organic letters, 20(21), 6794-6798 (2018-10-24)
A visible-light-mediated photocatalytic umpolung synthesis of 1,3-diamines from in situ-generated imines and dehydroalanine derivatives is described. Pivoting on a key nucleophilic addition of photocatalytically generated α-amino radicals to electron-deficient alkenes, this three-component coupling reaction affords 1,3-diamines efficiently and diastereoselectively. The
Timothy M Monos et al.
Science (New York, N.Y.), 361(6409), 1369-1373 (2018-09-29)
Alkene aminoarylation with a single, bifunctional reagent is a concise synthetic strategy. We report a catalytic protocol for the addition of arylsulfonylacetamides across electron-rich alkenes with complete anti-Markovnikov regioselectivity and excellent diastereoselectivity to provide 2,2-diarylethylamines. In this process, single-electron alkene
John C Tellis et al.
Science (New York, N.Y.), 345(6195), 433-436 (2014-06-07)
The routine application of C(sp3)-hybridized nucleophiles in cross-coupling reactions remains an unsolved challenge in organic chemistry. The sluggish transmetalation rates observed for the preferred organoboron reagents in such transformations are a consequence of the two-electron mechanism underlying the standard catalytic
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service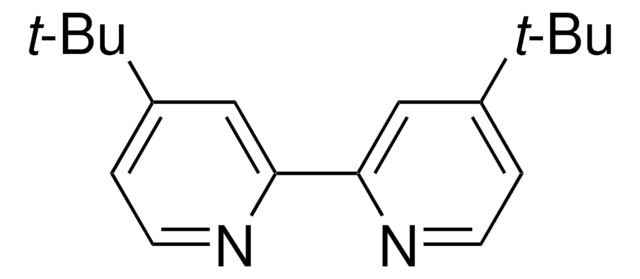
![(Ir[dF(CF3)ppy]2(dtbpy))PF6](/deepweb/assets/sigmaaldrich/product/structures/982/913/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09/640/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09.png)
![[Ir(dtbbpy)(ppy)2]PF6](/deepweb/assets/sigmaaldrich/product/structures/158/329/2544d673-d267-4aa1-8f46-2652aad4bfa0/640/2544d673-d267-4aa1-8f46-2652aad4bfa0.png)
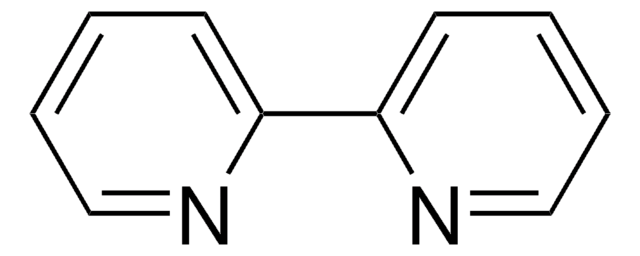
![[Ir{dFCF3ppy}2(bpy)]PF6](/deepweb/assets/sigmaaldrich/product/structures/180/924/79119ac4-7d62-429d-b23d-a14c012c6050/640/79119ac4-7d62-429d-b23d-a14c012c6050.png)