518751
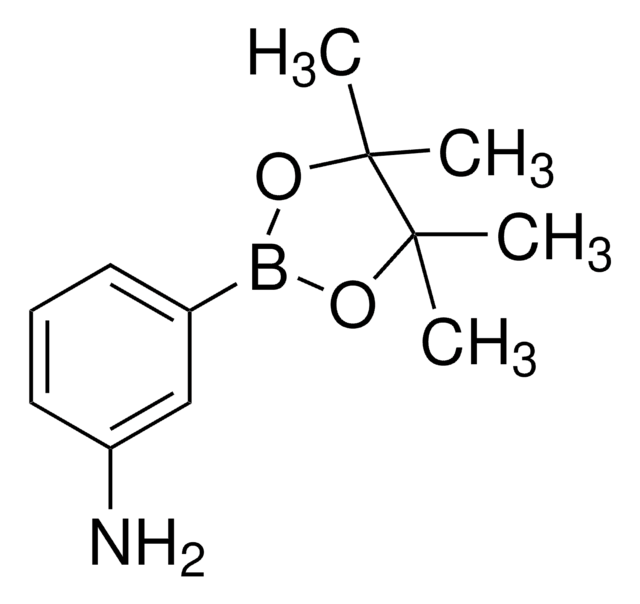
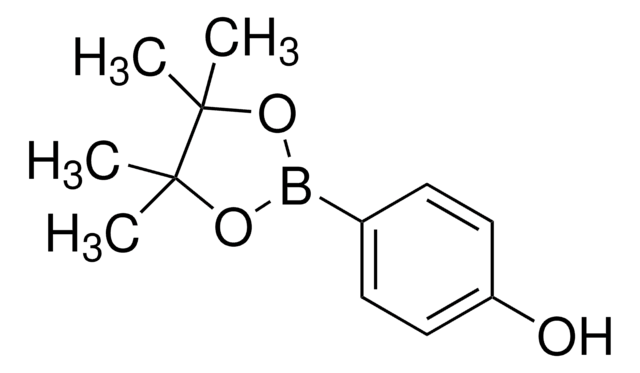
4-Aminophenylboronic acid pinacol ester
97%
Synonym(s):
2-(4-Aminophenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane, 4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)aniline, 4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)benzeneamine, 4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)phenylamine, 4-Aminophenylboronic acid, pinacol cyclic ester
About This Item
Recommended Products
Quality Level
Assay
97%
mp
165-169 °C (lit.)
SMILES string
CC1(C)OB(OC1(C)C)c2ccc(N)cc2
InChI
1S/C12H18BNO2/c1-11(2)12(3,4)16-13(15-11)9-5-7-10(14)8-6-9/h5-8H,14H2,1-4H3
InChI key
ZANPJXNYBVVNSD-UHFFFAOYSA-N
Related Categories
Application
- The preparation of substituted 3-phenyl-4H-1-benzopyran-4-ones by reacting with iodochromones via Pd catalyzed Suzuki-Miyaura cross-coupling reaction.
- Mercury(II) detection by fluorometry with new fluorogenic indicators based on through-bond energy transfer from pentaquinone to rhodamine.
- Rhodium-catalyzed amination reactions.
- Palladium-catalyzed Suzuki cross-coupling to synthesize potential antitubercular and antimicrobial compounds.
It can also be used to prepare:
- Hexaphenylbenzene derivatives as a potential bioprobe and multichannel keypad system.
- Pyromellitic diimide-based polymer as matrix for solution-processable n-channel field-effect transistors.
- Alternating copolymers of oligoarylenes and naphthalene bisimides as low band-gap semiconductors with electrochemical and spectroelectrochemical behavior.
- γ-secretase modulators in the treatment of amyloid β formation.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Arylboronic Acid-Pinacol Esters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service