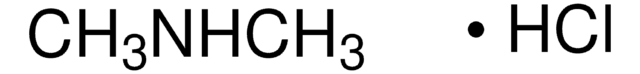426458
Dimethylamine solution
40 wt. % in H2O
Synonym(s):
DMA
About This Item
Recommended Products
vapor density
1.55 (vs air)
Quality Level
vapor pressure
16.97 psi ( 55 °C)
212 mmHg ( 20 °C)
form
liquid
autoignition temp.
753 °F
concentration
38-42% (with HCl, titration)
40 wt. % in H2O
refractive index
n20/D 1.37
density
0.89 g/mL at 25 °C
functional group
amine
SMILES string
CNC
InChI
1S/C2H7N/c1-3-2/h3H,1-2H3
InChI key
ROSDSFDQCJNGOL-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- To synthesize Ge7O14.5F2‚[(CH3)2NH2]3(H2O)0.86, a germanate-based porous inclusion complex.
- To synthesize ethyl 2-dimethylamino-4,6-diarylpyrimidine-5-carboxylates.
- To convert 5′-bromo-2,2′-bithiophene-5carboxaldehyde to 5′-dimethylamino-2,2′-bithiophene-5-carboxaldehyde.
related product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Inhalation - Aquatic Chronic 3 - Eye Dam. 1 - Flam. Liq. 2 - Skin Corr. 1B - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
3 - Flammable liquids
WGK
WGK 1
Flash Point(F)
60.8 °F - closed cup
Flash Point(C)
16 °C - closed cup
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 426458-100ML | 4061837673276 |
| 426458-1L | 4061832104300 |
| 426458-2L | 4061832104317 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service