所有图片(1)
About This Item
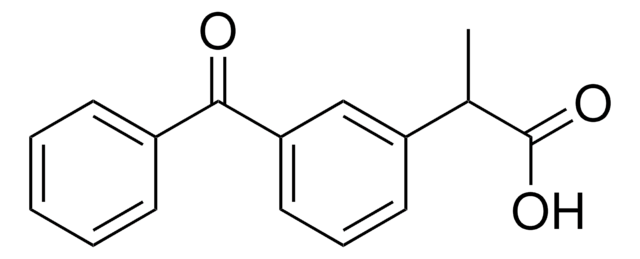
经验公式(希尔记法):
C16H14O3
CAS号:
分子量:
254.28
MDL號碼:
分類程式碼代碼:
41116107
PubChem物質ID:
NACRES:
NA.24
推荐产品
等級
pharmaceutical primary standard
API 家族
ketoprofen
製造商/商標名
USP
應用
pharmaceutical (small molecule)
形式
neat
SMILES 字串
CC(C(O)=O)c1cccc(c1)C(=O)c2ccccc2
InChI
1S/C16H14O3/c1-11(16(18)19)13-8-5-9-14(10-13)15(17)12-6-3-2-4-7-12/h2-11H,1H3,(H,18,19)
InChI 密鑰
DKYWVDODHFEZIM-UHFFFAOYSA-N
基因資訊
human ... PTGS1(5742) , PTGS2(5743)
正在寻找类似产品? 访问 产品对比指南
一般說明
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
應用
Ketoprofen USP reference standard, intended for use in specified quality tests and assays as specified in the USP compendia. Also, for use with USP monographs such as:
- Ketoprofen Capsules
- Ketoprofen Extended-Release Capsules
生化/生理作用
非甾体类抗炎化合物,具有COX-1选择性。
分析報告
These products are for test and assay use only. They are not meant for administration to humans or animals and cannot be used to diagnose, treat, or cure diseases of any kind.
其他說明
Sales restrictions may apply.
相關產品
产品编号
说明
价格
訊號詞
Danger
危險分類
Acute Tox. 3 Oral - Aquatic Acute 1 - Eye Irrit. 2 - Skin Irrit. 2
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 2
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
H Bagheri et al.
Drug safety, 22(5), 339-349 (2000-06-01)
The topical use of nonsteroidal anti-inflammatory drugs (NSAIDs), widely used for moderate acute and chronic painful conditions, is one of several strategies used to improve the tolerability profile of NSAIDs, particularly with regard to gastric and renal adverse effects. However
M Sugiura et al.
Contact dermatitis, 43(1), 16-19 (2000-07-21)
We report 4 cases of photocontact dermatitis due to ketoprofen, a non-steroidal anti-inflammatory drug derived from propionic acid. We carried out a skin biopsy to examine the amount of ketoprofen in the eruptive skin. We investigated the cross-reactions between tiaprofenic
Igor E Shohin et al.
Journal of pharmaceutical sciences, 101(10), 3593-3603 (2012-07-13)
Literature and experimental data relevant to the decision to allow a waiver of in vivo bioequivalence (BE) testing for the approval of immediate-release (IR) solid oral dosage forms containing ketoprofen are reviewed. Ketoprofen's solubility and permeability, its therapeutic use and
G Serrano et al.
Journal of the American Academy of Dermatology, 27(2 Pt 1), 204-208 (1992-08-01)
Photosensitivity reactions to fibric acid derivatives are not well understood and have been rarely reported. The aim of this study was to describe two cases of photosensitivity, one induced by fenofibrate and one by bezafibrate; to study the in vivo
Akihiko Ouchi et al.
Chirality, 14(1), 1-11 (2001-12-19)
The reactivity of organic molecules can be modified upon complexation with proteins: these changes can be different and more significant when the substrate is in an electronically excited state. Here we review UV, CD, and fluorescence spectroscopy studies on the
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门