推荐产品
品質等級
化驗
≥98% (HPLC)
形狀
powder
光學活性
[α]/D +80 to +95°, c = 1 (CHCl3)
顏色
white to beige
溶解度
DMSO: >5 mg/mL
儲存溫度
−20°C
SMILES 字串
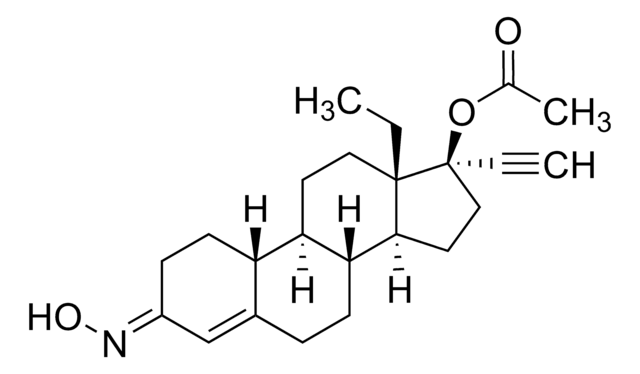
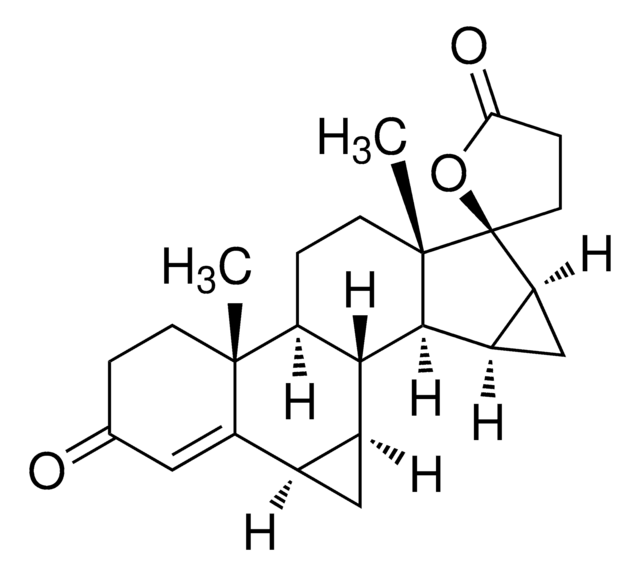
CC[C@]12CC(=C)[C@H]3[C@@H](CCC4=CC(=O)CC[C@H]34)[C@@H]1CC[C@@]2(O)C#C
InChI
1S/C22H28O2/c1-4-21-13-14(3)20-17-9-7-16(23)12-15(17)6-8-18(20)19(21)10-11-22(21,24)5-2/h2,12,17-20,24H,3-4,6-11,13H2,1H3/t17-,18-,19-,20+,21-,22-/m0/s1
InChI 密鑰
GCKFUYQCUCGESZ-BPIQYHPVSA-N
基因資訊
human ... PGR(5241)
正在寻找类似产品? 访问 产品对比指南
生化/生理作用
Etonogestrel is a lipophilic molecule. Pharmacokinetics studies reveal that etonogestrel is bound to protein albumin in the blood. This remains independent of both endogenous and exogenous concentration of estradiol.
Etonogestrel is a progestin, contraceptive.
Etonogestrel is a third-generation progestin contraceptive. Etonogestrel is used as a contraceptive itself and is also the active metabolite of the drug desogestrel used in some combination contraceptives.
特點和優勢
This compound is featured on the Nuclear Receptors (Steroids) page of the Handbook of Receptor Classification and Signal Transduction. To browse other handbook pages, click here.
訊號詞
Danger
危險聲明
危險分類
Carc. 2 - Repr. 1B
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
Pharmacokinetics of etonogestrel released from the contraceptive implant Implanon
Wenzl R, et al.
Contraception, 58(5), 283-288 (1998)
Peter Schnabel et al.
Clinical drug investigation, 32(6), 413-422 (2012-05-01)
The etonogestrel (ENG)-releasing implant is a subdermal progestogen-only contraceptive that provides coverage for up to 3 years. This long-acting hormonal contraceptive has been available in Europe since 1998 and in the US since 2006. To date, localization of non-palpable implants
Kristina M Tocce et al.
American journal of obstetrics and gynecology, 206(6), 481-481 (2012-05-29)
The purpose of this study was to determine contraceptive continuation and repeat pregnancy rates in adolescents who are offered immediate postpartum etonogestrel implant (IPI) insertion. Participants in an adolescent prenatal-postnatal program were enrolled in a prospective observational study of IPI
Diana Mansour et al.
Contraception, 83(3), 202-210 (2011-02-12)
The aim of this guidance is to review the management of unacceptable vaginal bleeding patterns in etonogestrel (ENG)-releasing contraceptive implant users concentrating, where possible, on the evidence for pharmacological treatments and identifying a pragmatic approach where this is not possible.
Anne Calhoun et al.
Headache, 52(8), 1246-1253 (2012-07-14)
To determine whether extended-cycle dosing of an ultralow dose vaginal ring contraceptive decreases frequency of migraine aura and prevents menstrual related migraine (MRM). Many women are denied therapy with combined hormonal contraceptives due to published guidelines that recommend against their
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门