推荐产品
生物源
human
重組細胞
expressed in HEK 293 cells
濃度
≥5000 units/mL
技術
protein extraction: suitable
適合性
suitable for additive or modifier in the separation of proteins or peptides
應用
life science and biopharma
運輸包裝
dry ice
儲存溫度
−20°C
一般說明
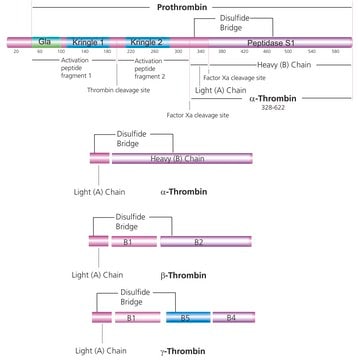
Thrombin is an endolytic serine protease that selectively cleaves the Arg–Gly bonds of fibrinogen to form fibrin and release fibrinopeptides A and B.1,2
The optimal cleavage sites for thrombin are as follows:
1. A-B-Pro-Arg-||-X-Y, where A and B are hydrophobic amino acids, and X and Y are nonacidic amino acids.
2. Gly-Arg-||-Gly
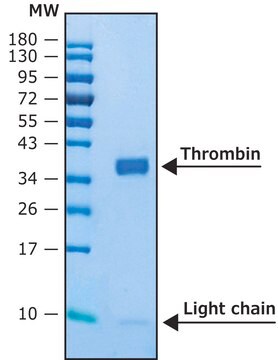
Recombinant human thrombin protease is expressed in human HEK 293 cells as a glycoprotein heterodimer. The DTT-reduced protein migrates as two bands of ∼31 kDa (heavy chain) and ∼6 kDa (light chain) on SDS-PAGE. This protein is manufactured in human cells, with no serum. The human cells expression system allows human-like glycosylation and folding, and often supports higher activity of the protein.
This thrombin protease is useful for cleaving recombinant proteins that are expressed as fusion proteins with this sequence between the carrier domain and the protein of interest.
The optimal cleavage sites for thrombin are as follows:
1. A-B-Pro-Arg-||-X-Y, where A and B are hydrophobic amino acids, and X and Y are nonacidic amino acids.
2. Gly-Arg-||-Gly
Recombinant human thrombin protease is expressed in human HEK 293 cells as a glycoprotein heterodimer. The DTT-reduced protein migrates as two bands of ∼31 kDa (heavy chain) and ∼6 kDa (light chain) on SDS-PAGE. This protein is manufactured in human cells, with no serum. The human cells expression system allows human-like glycosylation and folding, and often supports higher activity of the protein.
This thrombin protease is useful for cleaving recombinant proteins that are expressed as fusion proteins with this sequence between the carrier domain and the protein of interest.
應用
This biotinylated thrombin protease can be used for on-column cleavage of fusion proteins with a thrombin cleavage site. It specifically cleaves the protein of interest from a column-bound fusion protein, leaving the fusion domain or tag bound to the affinity column (e.g., Ni-NTA column) and eluting only the protein of interest.
This method is advantageous over post-elution cleavage for several reasons:
This method is advantageous over post-elution cleavage for several reasons:
- It eliminates most impurities normally associated with purification on Ni-chelating columns.
- It allows gentler elution conditions, with added flexibility in the elution buffer composition. This can mitigate protein aggregation and inactivation.
特點和優勢
The product is supplied in an aqueous buffer of pH 6.0, with 50% (v/v) glycerol.
Catalytic pH range:11.5–10
Optimal pH:11-8.3
(Note: thrombin precipitates at pH ≤5)
Molecular mass:4,12 37.4 kDa
Human isozymes pI range:6.35–7.6
E280 (1%):12 -18.3
Catalytic pH range:11.5–10
Optimal pH:11-8.3
(Note: thrombin precipitates at pH ≤5)
Molecular mass:4,12 37.4 kDa
Human isozymes pI range:6.35–7.6
E280 (1%):12 -18.3
This Thrombin protease, biotin-tagged enables on-column cleavage of fusion proteins with a thrombin cleavage site.
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 1
商品
Proteases for biotinylated tag removal for protein purification workflows with related reagents and technical resources.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门