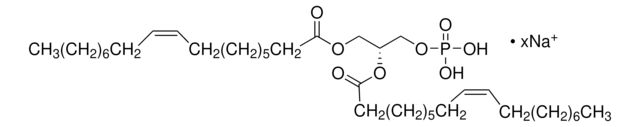
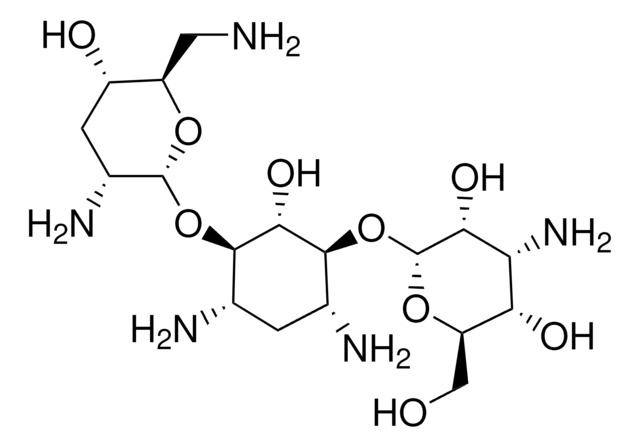
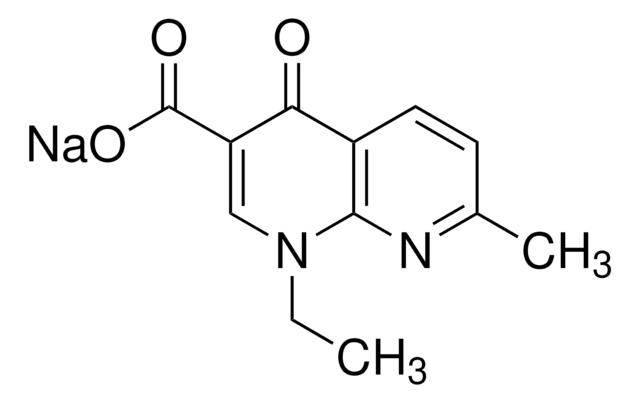
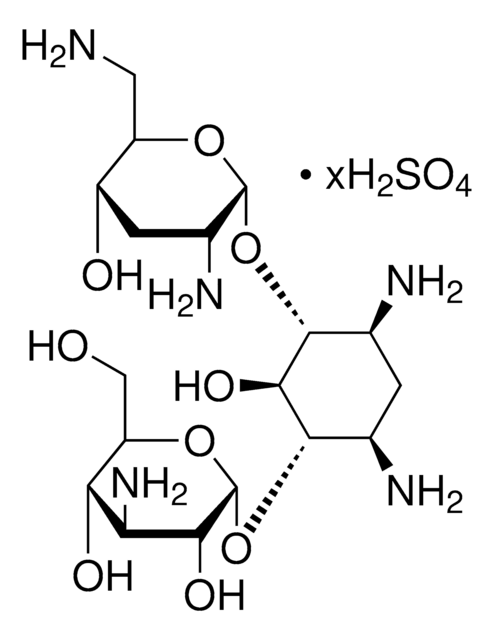
推荐产品
品質等級
化驗
≥98% (TLC)
形狀
powder
抗生素活性譜
Gram-negative bacteria
Gram-positive bacteria
parasites
作用方式
protein synthesis | interferes
SMILES 字串
O[C@H]1[C@H](O)[C@@H](CO)O[C@H](O[C@@]2([H])[C@H](O[C@@]3([H])[C@H](O)[C@H](O[C@]4([H])[C@H](N)[C@@H](O)[C@H](O)[C@H](CN)O4)[C@@H](CO)O3)[C@@H](O)[C@H](N)C[C@@H]2N)[C@@H]1N.C
InChI
1S/C23H45N5O14.CH4/c24-2-7-13(32)15(34)10(27)21(37-7)41-19-9(4-30)39-23(17(19)36)42-20-12(31)5(25)1-6(26)18(20)40-22-11(28)16(35)14(33)8(3-29)38-22;/h5-23,29-36H,1-4,24-28H2;1H4/t5-,6+,7+,8-,9-,10-,11-,12+,13-,14-,15-,16-,17-,18-,19-,20-,21-,22-,23+;/m1./s1
InChI 密鑰
OYJABWUHUYVDMJ-UDXJMMFXSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
Chemical structure: aminoglycoside
應用
Paromomycin is a broad spectrum aminoglycoside antibiotic produced by Streptomyces rimosus var. paromomycinus. It has in vitro and in vivo activity similar to neomycin. It is effective against Gram-negative bacteria, Gram-positive bacteria, some protozoan species, and limited antihelminth.
It is used to study bacterial protein synthesis at the level of 16S ribosomal RNA and 30S ribosome assembly. Paromomycin is used to study cytosine-cytosine (CC) mismatch-containing RNA molecules and is used to inhibit Cryptosporidium infection of a human enterocyte cell line.
It is used to study bacterial protein synthesis at the level of 16S ribosomal RNA and 30S ribosome assembly. Paromomycin is used to study cytosine-cytosine (CC) mismatch-containing RNA molecules and is used to inhibit Cryptosporidium infection of a human enterocyte cell line.
生化/生理作用
Paromomycin inhibits the initiation and elongation steps of protein synthesis by binding to 16S ribosomal RNA. Paramomycin binds to the A site, which causes defective polypeptide chains to be produced and leads to cell death.
包裝
1g,5g,25g
其他說明
Keep container tightly closed in a dry and well-ventilated place.
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
R J Marshall et al.
The Journal of infectious diseases, 165(4), 772-774 (1992-04-01)
Cryptosporidium parvum is a protozoan parasite that causes severe enteritis in patients with AIDS for which there is no effective therapy. Paromomycin is a nonabsorbable aminoglycoside that is effective in the treatment of other intestinal protozoa. The ability of paromomycin
Tony J Tavares et al.
RNA (New York, N.Y.), 15(5), 911-922 (2009-03-31)
The structure of a cytosine-cytosine (CC) mismatch-containing RNA molecule derived from a hairpin structure in the thymidylate synthase mRNA that binds the aminoglycoside paromomycin with high affinity was determined using nuclear magnetic resonance (NMR) spectroscopy. The cytosines in the mismatch
Kazuki Saito et al.
Nucleic acids research, 43(9), 4591-4601 (2015-04-22)
In eukaryotes, the tRNA-mimicking polypeptide-chain release factor, eRF1, decodes stop codons on the ribosome in a complex with eRF3; this complex exhibits striking structural similarity to the tRNA-eEF1A-GTP complex. Although amino acid residues or motifs of eRF1 that are critical
Sarah Hendrickx et al.
PLoS neglected tropical diseases, 6(5), e1664-e1664 (2012-06-06)
Paromomycin (PMM) has recently been introduced for treatment of visceral leishmaniasis in India. Although no clinical resistance has yet been reported, proactive vigilance should be warranted. The present in vitro study compared the outcome and stability of experimental PMM-resistance induction
Joline Goossens et al.
Toxins, 4(4), 281-295 (2012-05-19)
It is recognized that mycotoxins can cause a variety of adverse health effects in animals, including altered gastrointestinal barrier function. It is the aim of the present study to determine whether mycotoxin-contaminated diets can alter the oral bioavailability of the
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门