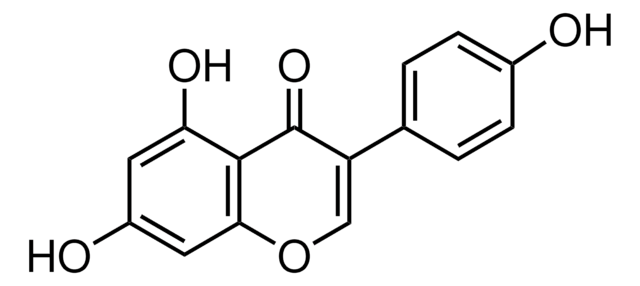
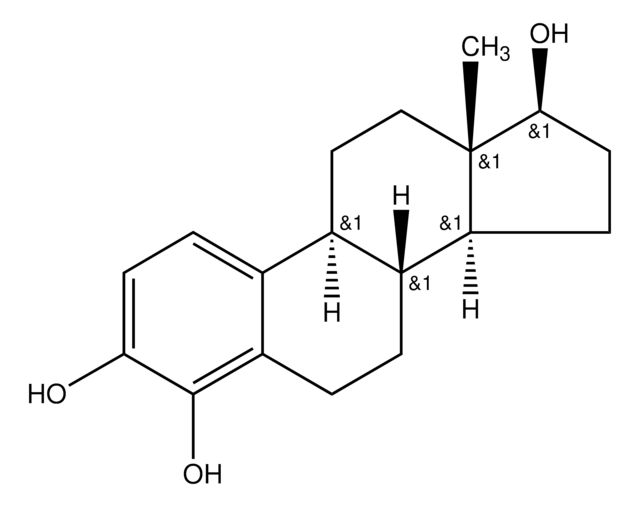
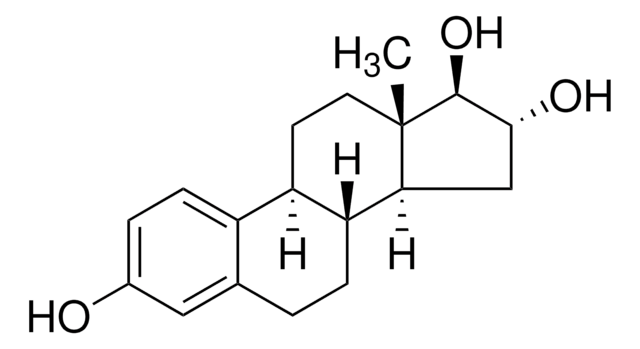
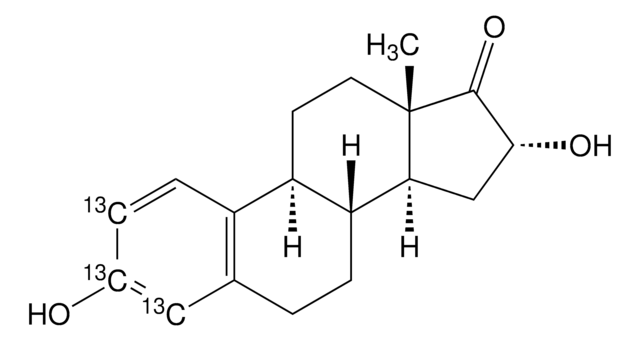
推荐产品
無菌
non-sterile
化驗
≥90% (HPLC)
形狀
powder
溶解度
methanol: 10 mg/mL, clear, colorless to faintly brownish-yellow
運輸包裝
ambient
儲存溫度
−20°C
SMILES 字串
[H][C@]12CC[C@]3(C)C(=O)CC[C@@]3([H])[C@]1([H])CCc4c(O)c(O)ccc24
InChI
1S/C18H22O3/c1-18-9-8-11-10-4-6-15(19)17(21)13(10)3-2-12(11)14(18)5-7-16(18)20/h4,6,11-12,14,19,21H,2-3,5,7-9H2,1H3/t11-,12-,14+,18+/m1/s1
InChI 密鑰
XQZVQQZZOVBNLU-QDTBLXIISA-N
生化/生理作用
4-Hydroxyestrone is an endogenous estrogen metabolite, which exhibits a strong neuroprotective effect against oxidative damage. It also provides effective protection against kanic acid-induced hippocampal oxidative damage in rats when compared to 17β-estradiol. 4-Hydroxyestrone regulates the angiogenic process during corpus luteum formation. It might be involved in an increased risk of cancer. 4-Hydroxyestrone is found in the early and mid-luteal phases.
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
199.9 °F - closed cup
閃點(°C)
93.3 °C - closed cup
個人防護裝備
Eyeshields, Gloves, type N95 (US)
Shelley A Gestl et al.
The American journal of pathology, 160(4), 1467-1479 (2002-04-12)
Glucuronidation, mediated by UDP-glucuronosyltransferases (UGTs), affects the actions and disposition of diverse endo- and xenobiotics. In the case of catecholestrogens (CEs), glucuronidation is likely to block their oxidation to quinone estrogens that are the putative mediators of CEs' actions as
Y Chen et al.
Chemical research in toxicology, 13(5), 342-350 (2000-05-17)
Estrogen replacement therapy has been correlated with an increased risk of developing breast or endometrial cancer. 4-Hydroxyequilenin (4-OHEN) is a catechol metabolite of equilenin which is a minor component of the estrogen replacement formulation marketed under the name of Premarin
B T Zhu et al.
Endocrinology, 135(5), 1772-1779 (1994-11-01)
As part of an ongoing investigation of the role of metabolic activation of estrogens in the genesis of cancers such as estrogen-induced renal tumors in hamsters, we have 1) determined steroid-17 beta-oxidoreductase activity of microsomes and cytosol prepared from hamster
X Xu et al.
The Journal of clinical endocrinology and metabolism, 84(11), 3914-3918 (1999-11-24)
Endogenous estrogen metabolism may play an important role in the pathogenesis of hormone-related cancers, most notably breast cancer. Despite the importance of estrogen metabolism, little is known about estrogen metabolite profiles during different phases of the menstrual cycle. This study
E Pisha et al.
Chemical research in toxicology, 14(1), 82-90 (2001-02-15)
Estrogen replacement therapy has been correlated with an increased risk of developing hormone-dependent cancers. 4-Hydroxyequilenin (4-OHEN) is a catechol metabolite of equilenin and equilin which are components of the estrogen replacement formulation marketed under the name of Premarin (Wyeth-Ayerst). Previously
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门