所有图片(1)
About This Item
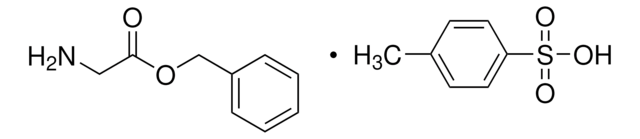
经验公式(希尔记法):
C9H11NO2 · HCl
CAS号:
分子量:
201.65
MDL號碼:
分類程式碼代碼:
12352209
eCl@ss:
32160406
PubChem物質ID:
NACRES:
NA.26
推荐产品
product name
甘氨酸苄酯 盐酸盐,
化驗
≥98% (TLC)
品質等級
形狀
powder
顏色
white
儲存溫度
−20°C
SMILES 字串
Cl.NCC(=O)OCc1ccccc1
InChI
1S/C9H11NO2.ClH/c10-6-9(11)12-7-8-4-2-1-3-5-8;/h1-5H,6-7,10H2;1H
InChI 密鑰
VLQHNAMRWPQWNK-UHFFFAOYSA-N
相关类别
應用
甘氨酸苄基酯可在缩合反应中用作有机试剂。
生化/生理作用
氨基酸单体的苯甲酯基团提高酶的底物亲和力和专一性,从而促进多肽的动力学控制合成(KCS)。它可用于甘氨酸单体的酶促化学聚合反应。
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
其他客户在看
J Martinez et al.
Carbohydrate research, 50(1), 15-22 (1976-08-01)
An O-glycodipeptide was synthesized by lengthening the peptide chain on the C-terminal side of a glycosylamino acid unit. N-(Benzyloxycarbonyl)-3-O-(2,3,4,6-tetra-O-acetyl-beta-D-glucopyranosyl)-L-threonine o-nitrophenyl ester and pentachlorophenyl ester were condensed with glycine benzyl ester to give both the same glycodipeptide, [N-(benzyloxycarbonyl)-3-O-(2,3,4,6-tetra-O-acetyl-beta-D-glucopyranosyl)-L-threonyl]glycine benzyl ester (9).
V Maurich et al.
Farmaco (Societa chimica italiana : 1989), 49(12), 805-808 (1994-12-01)
The synthesis of 2-(4-amino-5-chloro-2-methoxybenzamido)acetic acid 2, a metabolite of metoclopramide 1, has been accomplished through the coupling of 4-amino-5-chloro-2-methoxybenzoic acid 4 with glycine benzyl ester followed by a catalytic hydrogenation. Such a metabolite could not be detected directly in the
Jose Manuel Ageitos et al.
Biomacromolecules, 17(1), 314-323 (2015-12-02)
The chemoenzymatic polymerization of amino acid monomers by proteases involves a two-step reaction: the formation of a covalent acyl-intermediate complex between the protease and the carboxyl ester group of the monomer and the subsequent deacylation of the complex by aminolysis
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门