推荐产品
品質等級
化驗
≥98% (HPLC)
形狀
solid
顏色
white
溶解度
DMSO: >5 mg/mL
儲存溫度
−20°C
SMILES 字串
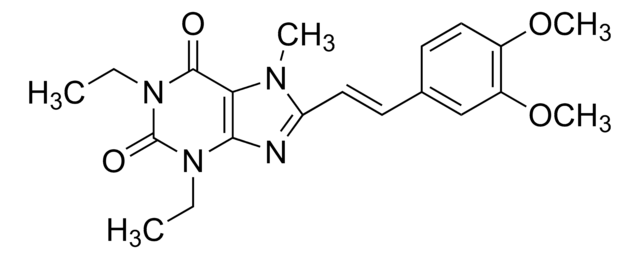
[H]\C(=C(\[H])c1nc2C(=O)N(C)C(=O)N(C)c2n1C)c3cccc(Cl)c3
InChI
1S/C16H15ClN4O2/c1-19-12(8-7-10-5-4-6-11(17)9-10)18-13-14(19)20(2)16(23)21(3)15(13)22/h4-9H,1-3H3/b8-7+
InChI 密鑰
MHYRUZOJQQLLQS-BQYQJAHWSA-N
基因資訊
human ... ADORA2A(135)
mouse ... Maob(109731)
rat ... Adora1(29290) , Adora2a(25369)
應用
8-(3-Chlorostyryl)caffeine has been used as an adenosine A2A receptor antagonist:
- to study its effects on levodopa (L-DOPA)-induced dopamine (DA) release in rat striatum
- to study its effect on the nuclear factor κ-light-chain-enhancer of activated B cells (NF-κB) activity in macrophages
- to determine its effects on human umbilical vein endothelial cells (HUVECs)
生化/生理作用
8-(3-Chlorostyryl)caffeine is a selective adenosine A2A receptor antagonist and monoamine oxidase B (MAO B) inhibitor.
特點和優勢
This compound is featured on the Adenosine Receptors page of the Handbook of Receptor Classification and Signal Transduction. To browse other handbook pages, click here.
注意
Photosensitive, store in the dark
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
Rangel L Silva et al.
Toxicology and applied pharmacology, 368, 63-71 (2019-02-24)
Cannabidiol (CBD) is a natural compound with psychoactive therapeutic properties well described. Conversely, the immunological effects of CBD are still poorly explored. In this study, the potential anti-inflammatory effects and underlying mechanisms of CBD and its analog Dimethyl-Heptyl-Cannabidiol (DMH-CBD) were
K A Jacobson et al.
Journal of medicinal chemistry, 36(10), 1333-1342 (1993-05-14)
A series of substituted 8-styryl derivatives of 1,3,7-alkylxanthines was synthesized as potential A2-selective adenosine receptor antagonists, and the potency at rat brain A1- and A2-receptors was studied in radioligand binding experiments. At the xanthine 7-position, only small hydrophobic substituents were
K A Jacobson et al.
FEBS letters, 323(1-2), 141-144 (1993-05-24)
An adenosine antagonist, 8-(3-chlorostyryl)caffeine (CSC), was shown previously to be 520-fold selective for A2a-adenosine receptors in radioligand binding assays in the rat brain. In reversing agonist effects on adenylate cyclase, CSC was 22-fold selective for A2a receptors in rat phenochromocytoma
Krystyna Gołembiowska et al.
Brain research, 998(2), 208-217 (2004-01-31)
In the present study, we investigated effects of the new selective adenosine A2A receptor antagonist 8-(3-chlorostyryl)caffeine (CSC) on L-DOPA-induced dopamine (DA) release in the striatum of intact and reserpine-treated rats. CSC given in a pharmacologically effective dose of 5 mg/kg
Eduardo P Amaral et al.
The Journal of infectious diseases, 219(6), 964-974 (2018-10-12)
Tuberculous pneumonia, necrotic granulomatous lesions, and bacterial dissemination characterize severe forms of mycobacterial infection. To evaluate the pulmonary CD4+ T-cell response during severe tuberculosis, C57BL/6 mice were infected with approximately 100 bacilli of 3 hypervirulent mycobacterial isolates (Mycobacterium tuberculosis strain
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门