推荐产品
等級
pharmaceutical primary standard
API 家族
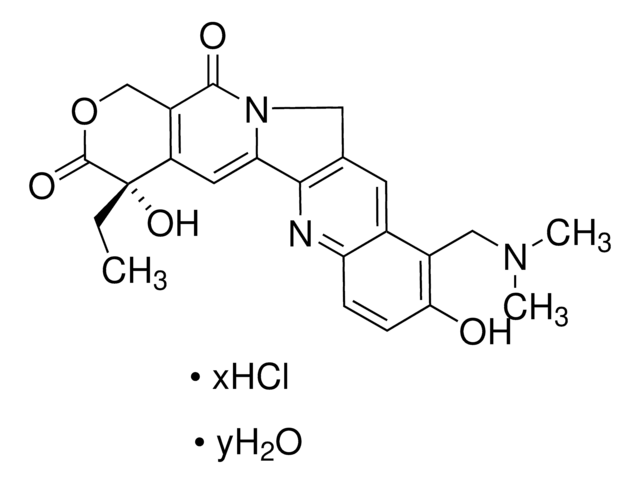
capecitabine
製造商/商標名
EDQM
應用
pharmaceutical (small molecule)
形式
neat
儲存溫度
2-8°C
SMILES 字串
O[C@H]1[C@@H](O)[C@H](N2C(N=C(NC(OCCCCC)=O)C(F)=C2)=O)O[C@@H]1C
InChI
1S/C15H22FN3O6/c1-3-4-5-6-24-15(23)18-12-9(16)7-19(14(22)17-12)13-11(21)10(20)8(2)25-13/h7-8,10-11,13,20-21H,3-6H2,1-2H3,(H,17,18,22,23)/t8-,10-,11-,13-/m1/s1
InChI 密鑰
GAGWJHPBXLXJQN-UORFTKCHSA-N
基因資訊
human ... TYMS(7298)
正在寻找类似产品? 访问 产品对比指南
一般說明
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
應用
Capecitabine EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
生化/生理作用
卡培他滨是一种抗癌药物,是去氧氟尿苷的前药,在肿瘤部位代谢为5-氟尿嘧啶。
卡培他滨是一种抗癌药物,是去氧氟尿苷的前药,在肿瘤部位代谢为5-氟尿嘧啶。 卡培他滨的激活遵循具有三个酶促步骤和两个中间代谢物5′-脱氧-5-氟胞苷(5′-DFCR)和5′-脱氧-5-氟尿苷(5′-DFUR)的途径,形成5-氟尿嘧啶。
包裝
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
其他說明
Sales restrictions may apply.
相關產品
产品编号
说明
价格
訊號詞
Danger
危險聲明
危險分類
Carc. 1B - Muta. 2 - Repr. 1B
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
Francesco Giotta et al.
Tumori, 99(6), 278e-281e (2014-02-08)
We present the case of a 58-year-old woman with breast cancer metastasizing to the liver after adjuvant chemotherapy. A liver biopsy confirmed metastatic lesions from breast cancer that were immunohistochemically positive for estrogen/progesterone receptors and HER2. After first-line treatment with
Karen-Lise G Spindler et al.
Anticancer research, 34(2), 845-850 (2014-02-11)
We investigated the efficacy and safety of capecitabine and gemcitabin (GemCap) in heavily pre-treated, therapy-resistant metastatic colorectal cancer (mCRC) patients and the clinical importance of cell-free DNA (cfDNA) measurement. Patients' inclusion criteria included histopathologically-verified mCRC refractory to standard chemotherapy, adequate
Dermatomyositis associated with capecitabine in the setting of malignancy.
Frank W Chen et al.
Journal of the American Academy of Dermatology, 70(2), e47-e48 (2014-01-21)
Mitsuhiro Tomoda et al.
Anticancer research, 34(1), 191-194 (2014-01-10)
Unresectable metastatic colorectal cancer with very slow tumour growth rate does not necessarily require for strong short-interval chemotherapy. In the present study, we administered monthly chemotherapy and aimed to evaluate the usefulness of the specific treatment schedule in patients with
Capecitabine and streptozocin ± cisplatin in advanced gastroenteropancreatic neuroendocrine tumours.
Tim Meyer et al.
European journal of cancer (Oxford, England : 1990), 50(5), 902-911 (2014-01-22)
Cytotoxic chemotherapy is widely used for advanced, unresectable pancreatic and other gastrointestinal foregut neuroendocrine tumours (NETs) and the most commonly used regimen combines 5-fluorouracil with streptozocin. The NET01 trial was designed to investigate whether capecitabine combined with streptozocin was an
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门