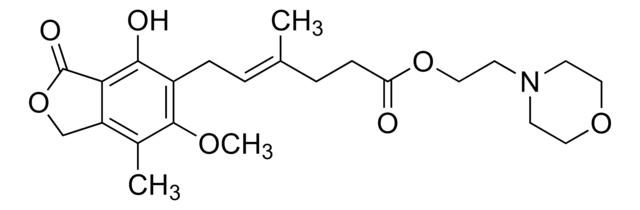
Y0000489
霉酚酸酯
European Pharmacopoeia (EP) Reference Standard
别名:
(4E)-6-(1,3-Dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexenoic acid 2-(4-morpholinyl)ethyl ester, RS 61443, TM-MMF
About This Item
推荐产品
等級
pharmaceutical primary standard
API 家族
mycophenolate
製造商/商標名
EDQM
應用
pharmaceutical (small molecule)
格式
neat
儲存溫度
2-8°C
SMILES 字串
COc1c(C)c2COC(=O)c2c(O)c1C\C=C(/C)CCC(=O)OCCN3CCOCC3
InChI
1S/C23H31NO7/c1-15(5-7-19(25)30-13-10-24-8-11-29-12-9-24)4-6-17-21(26)20-18(14-31-23(20)27)16(2)22(17)28-3/h4,26H,5-14H2,1-3H3/b15-4+
InChI 密鑰
RTGDFNSFWBGLEC-SYZQJQIISA-N
基因資訊
human ... IMPDH1(3614) , IMPDH2(3615)
正在寻找类似产品? 访问 产品对比指南
一般說明
應用
生化/生理作用
包裝
其他說明
相關產品
訊號詞
Danger
危險分類
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Repr. 1B - STOT RE 1
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门