推荐产品
重組細胞
expressed in mouse cell line
品質等級
化驗
≥90% (SDS-PAGE)
形狀
liquid
不包含
preservative
製造商/商標名
Calbiochem®
儲存條件
OK to freeze
avoid repeated freeze/thaw cycles
運輸包裝
wet ice
儲存溫度
−70°C
一般說明
Recombinant, human pro-MMP-2 expressed in mouse cells. Useful for immunoblotting, substrate cleavage and assay zymography. The ~72 kDa purified proenzyme MMP-2 may be used as a positive control or standard for zymographic analysis, immunoblotting, or substrate cleavage assays. Requires activation prior to use. Matrix metalloproteinases are members of a unique family of proteolytic enzymes that have a zinc ion at their active sites and can degrade collagens, elastin and other components of the extracellular matrix (ECM). These enzymes are present in normal healthy individuals and have been shown to have an important role in processes such as wound healing, pregnancy, and bone resorption. However, overexpression and activation of MMPs have been linked with a range of pathological processes and disease states involved in the breakdown and remodeling of the ECM. Such diseases include tumor invasion and metastasis, rheumatoid arthritis, periodontal disease and vascular processes such as angiogenesis, intimal hyperplasia, atherosclerosis and aneurysms. Recently, MMPs have been linked to neurodegenerative diseases such as Alzheimer′s, and amyotrophic lateral sclerosis (ALS). Natural inhibitors of MMPs, tissue inhibitor of matrix metalloproteinases (TIMPs) exist and synthetic inhibitors have been developed which offer hope of new treatment options for these diseases.
Regulation of MMP activity can occur at the level of gene expression, including transcription and translation, level of activation, or at the level of inhibition by TIMPs. Thus, perturbations at any of these points can theoretically lead to alterations in ECM turnover. Expression is under tight control by pro- and anti-inflammatory cytokines and/or growth factors and, once produced the enzymes are usually secreted as inactive zymograms. Upon activation (removal of the inhibitory propeptide region of the molecules) MMPs are subject to control by locally produced TIMPs1. All MMPs can be activated in vitro with organomercurial compounds (e.g., 4-aminophenylmercuric acetate), but the agents responsible for the physiological activation of all MMPs have not been clearly defined. Numerous studies indicate that members of the MMP family have the ability to activate one another13. The activation of the MMPs in vivo is likely to be a critical step in terms of their biological behavior, because it is this activation that will tip the balance in favor of ECM degradation. The hallmark of diseases involving MMPs appear to be stoichiometric imbalance between active MMPs and TIMPs, leading to excessive tissue disruption and often degradation. Determination of the mechanisms that control this imbalance may open up some important therapeutic options of specific enzyme inhibitors.
Regulation of MMP activity can occur at the level of gene expression, including transcription and translation, level of activation, or at the level of inhibition by TIMPs. Thus, perturbations at any of these points can theoretically lead to alterations in ECM turnover. Expression is under tight control by pro- and anti-inflammatory cytokines and/or growth factors and, once produced the enzymes are usually secreted as inactive zymograms. Upon activation (removal of the inhibitory propeptide region of the molecules) MMPs are subject to control by locally produced TIMPs1. All MMPs can be activated in vitro with organomercurial compounds (e.g., 4-aminophenylmercuric acetate), but the agents responsible for the physiological activation of all MMPs have not been clearly defined. Numerous studies indicate that members of the MMP family have the ability to activate one another13. The activation of the MMPs in vivo is likely to be a critical step in terms of their biological behavior, because it is this activation that will tip the balance in favor of ECM degradation. The hallmark of diseases involving MMPs appear to be stoichiometric imbalance between active MMPs and TIMPs, leading to excessive tissue disruption and often degradation. Determination of the mechanisms that control this imbalance may open up some important therapeutic options of specific enzyme inhibitors.
包裝
Please refer to vial label for lot-specific concentration.
警告
Toxicity: Standard Handling (A)
外觀
In 150 mM NaCl, 20 mM Tris-HCl, 5 mM CaCl₂, 0.05% BRIJ®-35 Detergent, pH 7.4.
重構
Following initial thaw, aliquot and freeze (-70°C).
分析報告
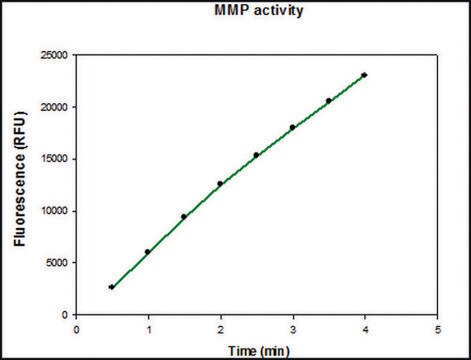
Specific Activity: Measured by its ability to degrade gelatin using zymography. 1 ng of enzyme is sufficient to visualize degraded gelatin by coomassie blue stain in zymography. Activated rhMMP-2 degrades type IV collagen, as visualized by SDS-PAGE and cleaves the coumarin peptide substrate, (7-methoxycoumarin-4-yl) Acetyl-Pro-Leu-Gly-Leu-(3-[2, 4-dinitrophenyl-L-2, 3-diaminopropionyl)-Ala-Arg-NH2 (Cat. No. 03-32-5032), as measured by fluorescence (excitation at 320 nm and emission at 405 nm).
其他說明
Buisson-Leqendre, N., et al. 2004. Arth. Rheumat.50, 2151.
Parsons, S.L., et al. 1997. Br. J. Surg.84, 160.
Backstrom, J.R., et al. 1996. J. Neuro.16, 7910.
Lim, G.P., et al. 1996. J. Neurochem.67, 251.
Xia, T., et al. 1996. Biochim. Biophys. Acta1293, 259.
Chandler, S. et al. 1995. Neurosci. Lett.201, 223.
Sang, Q.X., et al. 1995. Biochim. Biophys. Acta1251, 99.
Kenagy, R.D. and Clowes, A.W., 1994. In Inhibition of Matrix Metalloproteinases: Therapeutic Potential. Greenwald, R.A. and Golub L.M., Eds. 462.
Kleiner, D.E. and Stetler-Stevenson W.G., 1994. Anal. Biochem.218, 325.
Zempo, N., et al. 1994. J. Vasc. Surg.20, 209.
Birkedal-Hansen, H. 1993. J. Periodontol.64 474.
Stetler-Stevenson, W.G., et al. 1993. FASEB J.7, 1434.
Delaisse, J-M. and Vaes, G. 1992. In Biology and Physiology of the Osteoclast. B.R. Rifkin & C.V. Gay, Eds. 290.
Jeffrey, J.J. 1992. In Wound Healing: Biochemical and Clinical Aspects. R.F. Diegelmann and W.J. Lindblad, Eds. 177.
Jeffrey, J.J. 1991. Semin. Perinatol.15, 118.
Liotta, L.A., et al. 1991. Cell64, 327.
Harris, E. 1990. N. Engl. J. Med.322, 1277.
Heussen, C. and Dowdle, E.B., 1980. Anal. Biochem.102, 196.
Parsons, S.L., et al. 1997. Br. J. Surg.84, 160.
Backstrom, J.R., et al. 1996. J. Neuro.16, 7910.
Lim, G.P., et al. 1996. J. Neurochem.67, 251.
Xia, T., et al. 1996. Biochim. Biophys. Acta1293, 259.
Chandler, S. et al. 1995. Neurosci. Lett.201, 223.
Sang, Q.X., et al. 1995. Biochim. Biophys. Acta1251, 99.
Kenagy, R.D. and Clowes, A.W., 1994. In Inhibition of Matrix Metalloproteinases: Therapeutic Potential. Greenwald, R.A. and Golub L.M., Eds. 462.
Kleiner, D.E. and Stetler-Stevenson W.G., 1994. Anal. Biochem.218, 325.
Zempo, N., et al. 1994. J. Vasc. Surg.20, 209.
Birkedal-Hansen, H. 1993. J. Periodontol.64 474.
Stetler-Stevenson, W.G., et al. 1993. FASEB J.7, 1434.
Delaisse, J-M. and Vaes, G. 1992. In Biology and Physiology of the Osteoclast. B.R. Rifkin & C.V. Gay, Eds. 290.
Jeffrey, J.J. 1992. In Wound Healing: Biochemical and Clinical Aspects. R.F. Diegelmann and W.J. Lindblad, Eds. 177.
Jeffrey, J.J. 1991. Semin. Perinatol.15, 118.
Liotta, L.A., et al. 1991. Cell64, 327.
Harris, E. 1990. N. Engl. J. Med.322, 1277.
Heussen, C. and Dowdle, E.B., 1980. Anal. Biochem.102, 196.
法律資訊
Brij is a registered trademark of Croda International PLC
CALBIOCHEM is a registered trademark of Merck KGaA, Darmstadt, Germany
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 1
閃點(°F)
Not applicable
閃點(°C)
Not applicable
M T Kato et al.
Caries research, 44(3), 309-316 (2010-06-17)
It is known that some metal salts can inhibit matrix metalloproteinase (MMP) activity, but the effect of iron has not been tested yet. On the other hand, it has recently been suggested that MMP inhibition might influence dentine erosion. Based
Cintia Silveira et al.
Caries research, 48(6), 549-556 (2014-07-06)
The aim of this study was to analyze the effect of solutions containing saturated anacardic acid (AA) on dentine erosion in vitro. AA was chemically isolated from natural cashew nutshell liquid obtained by continuous extraction in a Soxhlet extractor and
Catalina Vallejo-Giraldo et al.
Frontiers in bioengineering and biotechnology, 8, 601704-601704 (2020-11-27)
The development of 3D neural tissue analogs is of great interest to a range of biomedical engineering applications including tissue engineering of neural interfaces, treatment of neurodegenerative diseases and in vitro assessment of cell-material interactions. Despite continued efforts to develop
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门