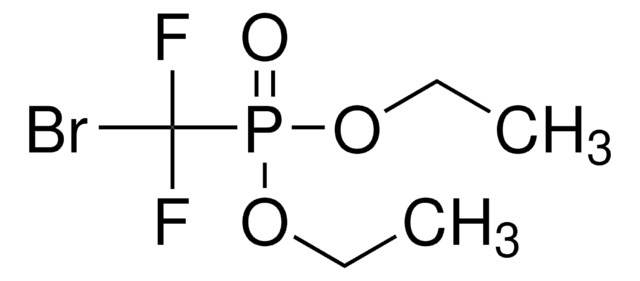
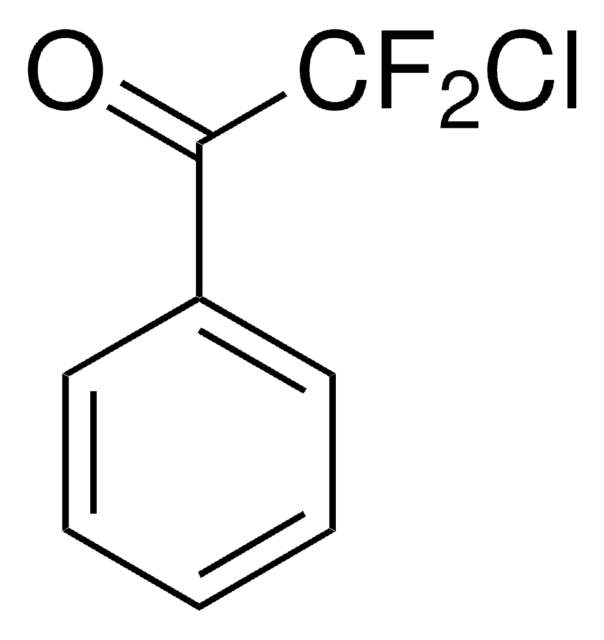
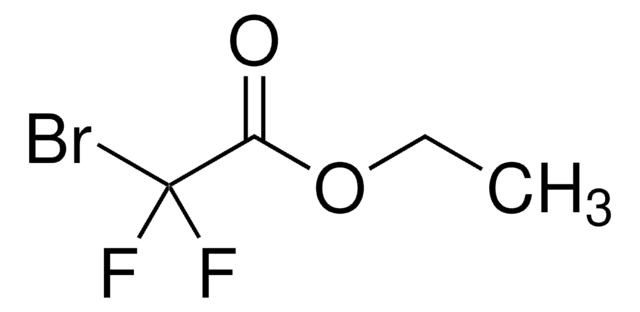
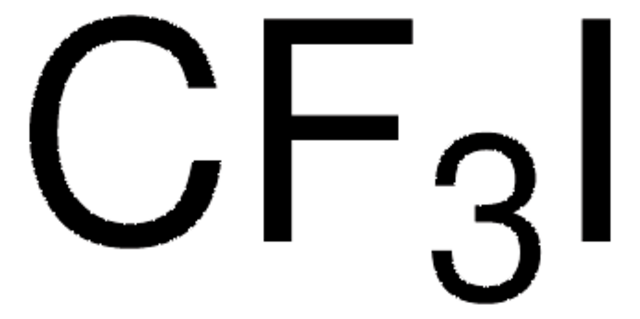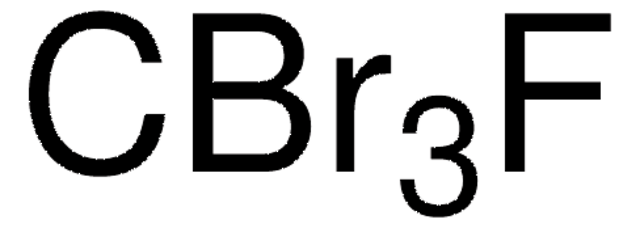推荐产品
化驗
≥95.0%
形狀
solid
儲存溫度
2-8°C
InChI
1S/C19H16F2P.BrH/c20-19(21)22(16-10-4-1-5-11-16,17-12-6-2-7-13-17)18-14-8-3-9-15-18;/h1-15,19H;1H/q+1;/p-1
InChI 密鑰
WNPMJTVOWUTTSY-UHFFFAOYSA-M
應用
When irradiated with visible light, difluoromethyltriphenylphosphonium bromide generates a difluoromethyl radical, which has been shown to react with alkenes, enamides, and thiols to give the difluoromethylated product.
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
相關產品
产品编号
说明
价格
訊號詞
Danger
危險分類
Acute Tox. 3 Oral - Acute Tox. 4 Dermal - Aquatic Chronic 2 - Eye Dam. 1 - Skin Irrit. 2
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 3
Qing-Yu Lin et al.
Angewandte Chemie (International ed. in English), 55(4), 1479-1483 (2015-12-17)
Bromodifluoromethylphosphonium bromide was solely used as the precursor of difluorocarbene. Herein, an unprecedented visible-light-induced hydrodifluoromethylation of alkenes with bromodifluoromethylphosphonium bromide using H2O and THF as hydrogen sources for the synthesis of difluoromethylated alkanes is described. This difluoromethylation is characterized by
Niklas B Heine et al.
Organic letters, 19(15), 4150-4153 (2017-07-21)
A method for facile difluoromethylation of various thiols using (difluoromethyl)triphenylphosphonium bromide under mild reaction conditions is presented. The transformation proceeds in the absence of any transition metal using a bench-stable and readily accessible phosphonium salt. Deuterium labeling experiments and cyclic
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门