所有图片(1)
About This Item
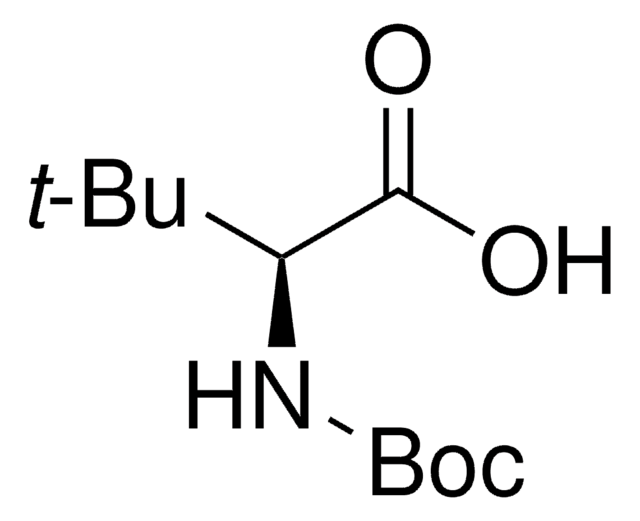
线性分子式:
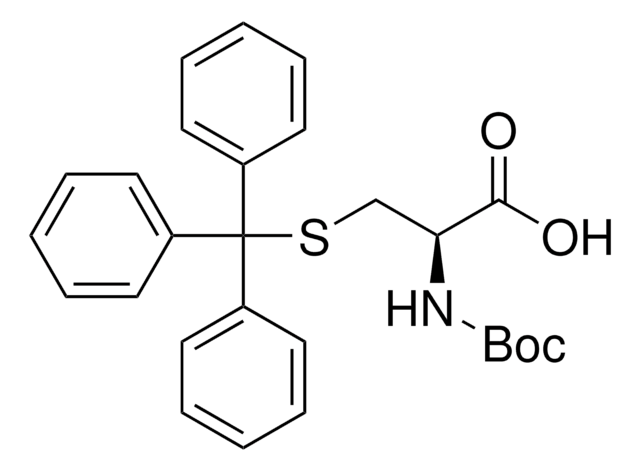
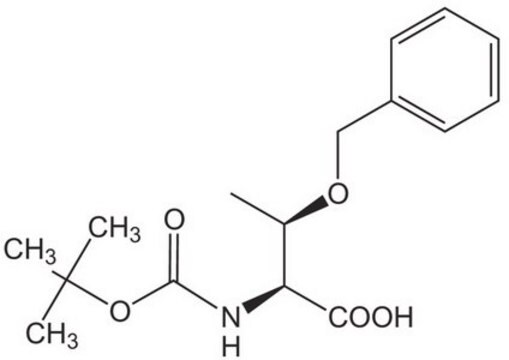
(CH3)3COCH(CH3)CH(COOH)NHCOOC(CH3)3
CAS号:
分子量:
275.34
Beilstein:
4454820
MDL號碼:
分類程式碼代碼:
12352209
PubChem物質ID:
NACRES:
NA.22
推荐产品
化驗
95%
品質等級
形狀
powder
mp
98.9 °C
應用
peptide synthesis
儲存溫度
2-8°C
SMILES 字串
C[C@@H](OC(C)(C)C)[C@H](NC(=O)OC(C)(C)C)C(O)=O
InChI
1S/C13H25NO5/c1-8(18-12(2,3)4)9(10(15)16)14-11(17)19-13(5,6)7/h8-9H,1-7H3,(H,14,17)(H,15,16)/t8-,9+/m1/s1
InChI 密鑰
LKRXXARJBFBMCE-BDAKNGLRSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
Boc-Thr(t-Bu)-OH (N-Boc-O-tert-butyl-L-threonine) participates in the synthesis of 2,3-unsaturated glycosides, via reaction with per-O-acetylated glucal in the presence of Er(OTf)3 catalyst.
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Antonio Procopio et al.
Carbohydrate research, 342(14), 2125-2131 (2007-06-23)
Er(OTf)(3) is a useful catalyst for the Ferrier rearrangement furnishing high yields of O- and S-glycosides. The transformation has wide applicability, cleaner reaction profiles, mild reaction conditions, and high stereoselectivity and the catalyst, which is also commercially available, can be
Christian Haenig et al.
Cell reports, 32(7), 108050-108050 (2020-08-20)
Interactome maps are valuable resources to elucidate protein function and disease mechanisms. Here, we report on an interactome map that focuses on neurodegenerative disease (ND), connects ∼5,000 human proteins via ∼30,000 candidate interactions and is generated by systematic yeast two-hybrid
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门