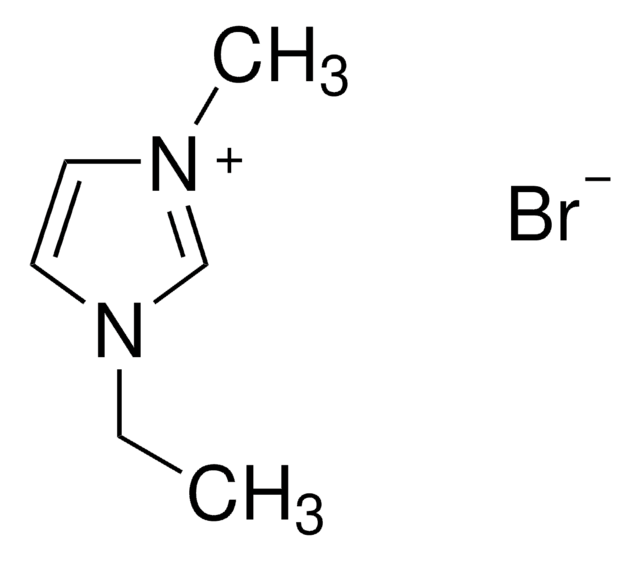
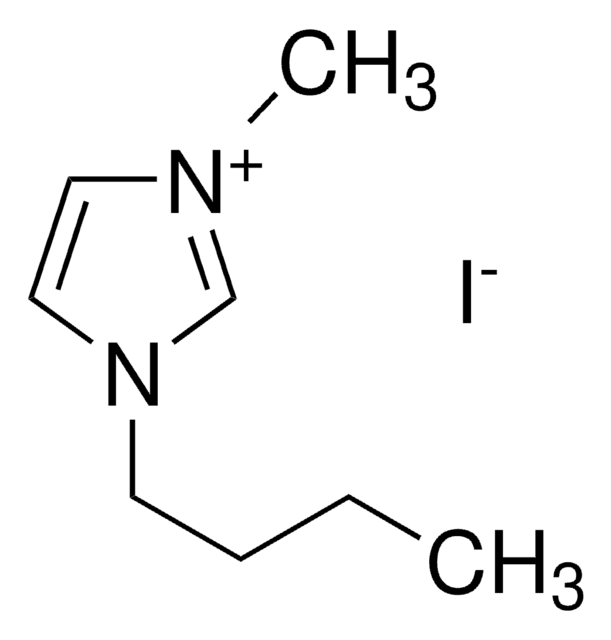
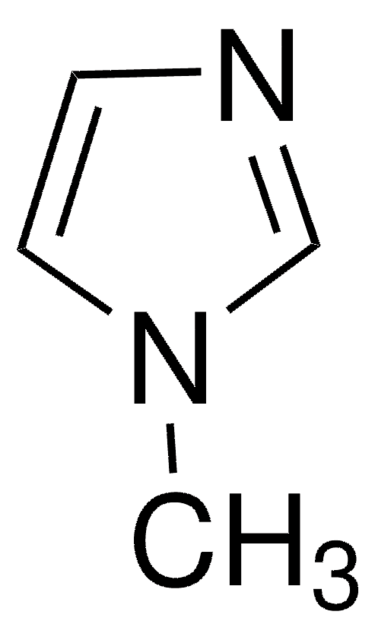
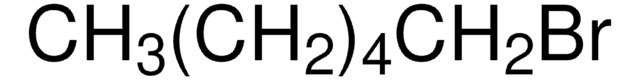About This Item
推荐产品
化驗
≥98.5% (HPLC)
形狀
solid
雜質
≤500 ppm water
SMILES 字串
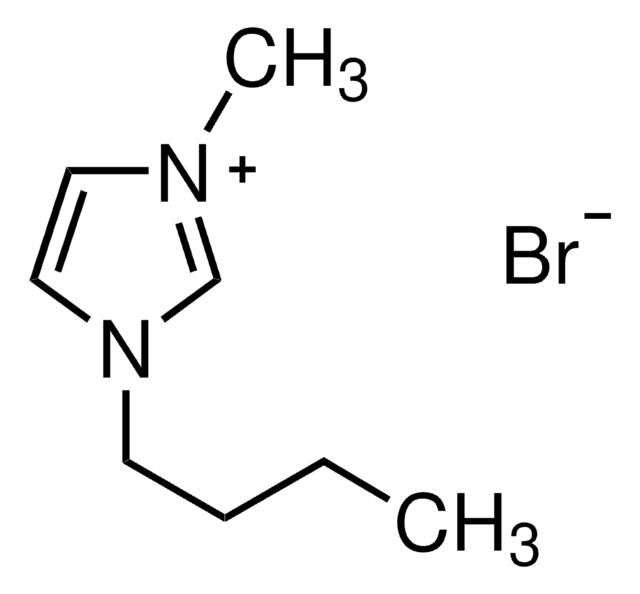
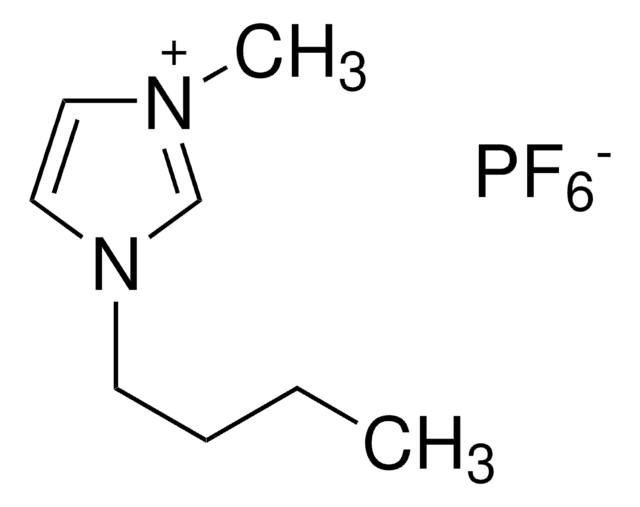
[Br-].CCCCn1cc[n+](C)c1
InChI
1S/C8H15N2.BrH/c1-3-4-5-10-7-6-9(2)8-10;/h6-8H,3-5H2,1-2H3;1H/q+1;/p-1
InChI 密鑰
KYCQOKLOSUBEJK-UHFFFAOYSA-M
正在寻找类似产品? 访问 产品对比指南
一般說明
應用
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
其他客户在看
商品
Ionic Liquids have been thoroughly investigated as solvents in most types of catalytic reactions. Their merit lies in the ease with which their physical–chemical properties can be tuned by varying either the anion, the cation, or its substitution pattern.
Ionic Liquids have been thoroughly investigated as solvents in most types of catalytic reactions. Their merit lies in the ease with which their physical–chemical properties can be tuned by varying either the anion, the cation, or its substitution pattern.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门