所有图片(1)
About This Item
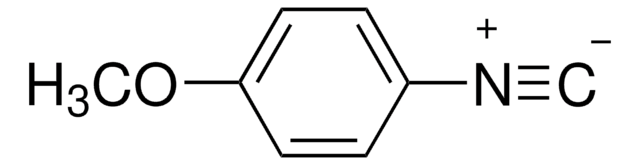
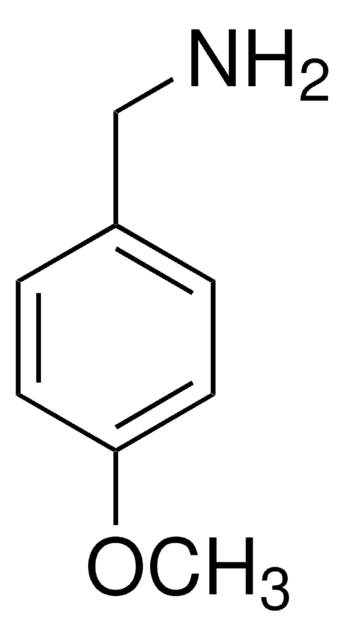
线性分子式:
CH3OC6H4CH2NCO
CAS号:
分子量:
163.17
MDL號碼:
分類程式碼代碼:
12352100
PubChem物質ID:
NACRES:
NA.22
推荐产品
化驗
98%
折射率
n20/D 1.433 (lit.)
bp
60-66 °C/0.6 mmHg (lit.)
密度
1.143 g/mL at 25 °C (lit.)
儲存溫度
2-8°C
SMILES 字串
COc1ccc(CN=C=O)cc1
InChI
1S/C9H9NO2/c1-12-9-4-2-8(3-5-9)6-10-7-11/h2-5H,6H2,1H3
InChI 密鑰
QRBHVARIMDDOOV-UHFFFAOYSA-N
一般說明
4-Methoxybenzyl isocyanate (PMBNCO), also known as p-methoxybenzyl isocyanate, is an organic building block containing an isocyanate group. Its enthalpy of vaporization at boiling point has been reported.
應用
4-Methoxybenzyl isocyanate (p-Methoxybenzyl isocyanate) may be used in the synthesis of 1-(4-methoxybenzyl)-3-{2-[(4-nitrophenyl)amino]ethyl}urea and 1-(4-methoxybenzyl)-3-{2-[(5-nitropyridin-2-yl)amino]ethyl} urea.
It may be used in the multi-step synthesis of:
It may be used in the multi-step synthesis of:
- diimidazodiazepine containing 5:7:5-fused heterocycle
- 2-deoxy-2-ureido-D-ribo-hexulofuranosonamide
- bicyclic guanidine core of batzelladine A
訊號詞
Danger
危險分類
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Resp. Sens. 1 - Skin Irrit. 2 - Skin Sens. 1 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 3
閃點(°F)
235.4 °F - closed cup
閃點(°C)
113 °C - closed cup
個人防護裝備
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Michael A Arnold et al.
Journal of the American Chemical Society, 128(40), 13255-13260 (2006-10-05)
A diastereoselective [4 + 2]-annulation of vinyl carbodiimides with chiral N-alkyl imines has been developed to access the stereochemically rich polycyclic guanidine cores of the batzelladine alkaloids. Application of this strategy, together with additional key steps such as long-range directed
Raj Kumar et al.
Organic letters, 10(20), 4681-4684 (2008-09-26)
The first synthesis of a novel 5:7:5-fused heterocyclic ring system, a diimidazodiazepine, is reported. The propensity of the ring system to undergo facile, acid-catalyzed nucleophilic addition reactions by neutral carbon and nitrogen nucleophiles has been explored. The ring system has
Synthesis and herbicidal activity of opened hydantoin-ring derivatives of hydantocidin.
Hiromi S, et al.
Bioscience, Biotechnology, and Biochemistry, 60(7), 1198-1200 (1996)
Tomasz M Wróbel et al.
Journal of enzyme inhibition and medicinal chemistry, 31(4), 608-618 (2015-06-27)
A series of urea derivatives bearing nitroaryl moiety has been synthesized and assayed for their potential antiproliferative activities. Some of the tested compounds displayed activity in RK33 laryngeal cancer cells and TE671 rhabdomyosarcoma cells while being generally less toxic to
Yaws CL.
Thermophysical Properties of Chemicals and Hydrocarbons, 567-567 (2014)
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门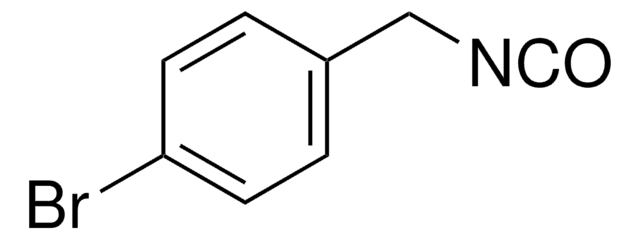

![(Ir[dF(CF3)ppy]2(dtbpy))PF6](/deepweb/assets/sigmaaldrich/product/structures/982/913/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09/640/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09.png)