所有图片(2)
About This Item
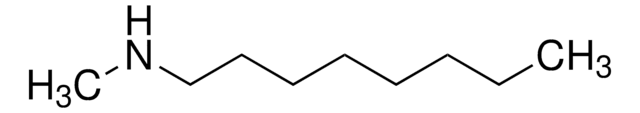
线性分子式:
CH3NH(CH2)5CH3
CAS号:
分子量:
115.22
Beilstein:
1731685
EC號碼:
MDL號碼:
分類程式碼代碼:
12352100
PubChem物質ID:
NACRES:
NA.22
推荐产品
品質等級
化驗
96%
形狀
liquid
折射率
n20/D 1.416 (lit.)
bp
140-142 °C (lit.)
密度
0.76 g/mL at 25 °C (lit.)
官能基
amine
SMILES 字串
CCCCCCNC
InChI
1S/C7H17N/c1-3-4-5-6-7-8-2/h8H,3-7H2,1-2H3
InChI 密鑰
XJINZNWPEQMMBV-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
N-Hexylmethylamine is an acyclic secondary amine. Ruthenium-catalyzed reaction of N-hexylmethylamine with styrene has been reported. Transition metal-catalyzed intermolecular hydroamination of N-hexylmethylamine with 2-vinylnaphthalene has been reported.
應用
N-Hexylmethylamine may be used in the synthesis of dialkyldithiocarbamato cadmium complexes Cd[S2CNRR′]2.
訊號詞
Danger
危險聲明
危險分類
Acute Tox. 4 Oral - Flam. Liq. 3 - Skin Corr. 1B
儲存類別代碼
3 - Flammable liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
87.8 °F - closed cup
閃點(°C)
31 °C - closed cup
個人防護裝備
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
其他客户在看
Masaru Utsunomiya et al.
Journal of the American Chemical Society, 126(9), 2702-2703 (2004-03-05)
A ruthenium-catalyzed intermolecular, anti-Markovnikov hydroamination of vinylarenes with secondary aliphatic and benzylic amines is reported. The combination of Ru(cod)(2-methylallyl)2, 1,5-bis(diphenylphosphino)pentane, and triflic acid was the most effective catalyst of those tested. Control reactions conducted without ligand or acid did not
The synthesis of SiO2@ CdS nanocomposites using single-molecule precursors.
Monteiro OC, et al.
Chemistry of Materials, 14(7), 2900-2904 (2002)
Masaru Utsunomiya et al.
Journal of the American Chemical Society, 125(47), 14286-14287 (2003-11-20)
A transition metal-catalyzed intermolecular hydroamination of vinylarenes with alkylamines is reported. The combination of Pd(O2CCF3)4, DPPF, and TfOH was the most effective catalyst of those tested. Control experiments without palladium, acid, or ligand all occurred in low yield. The reaction
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门