所有图片(1)
About This Item
线性分子式:
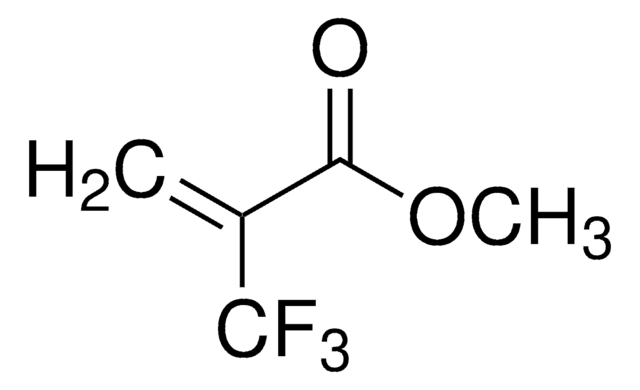
H2C=C(NHCOCH3)CO2CH3
CAS号:
分子量:
143.14
MDL號碼:
分類程式碼代碼:
12162002
PubChem物質ID:
NACRES:
NA.23
推荐产品
化驗
98%
bp
104 °C/8 mmHg (lit.)
mp
50-52 °C (lit.)
SMILES 字串
COC(=O)C(=C)NC(C)=O
InChI
1S/C6H9NO3/c1-4(6(9)10-3)7-5(2)8/h1H2,2-3H3,(H,7,8)
InChI 密鑰
SMWNFFKPVLVOQQ-UHFFFAOYSA-N
一般說明
Conjugated addition of secondary amines, imidazole and pyrazole to methyl 2 methyl 2-acetamidoacrylate in the presence of a catalyst results in the formation of β-Dialkylamino-α-alanine and β-(N-heteroaryl)-α-alanine derivatives. Methyl-2-acetamidoacrylate (M2AA) is an anti-inflammatory agent. The catalytic reaction of methyl 2-acetamidoacrylate with Grignard′s reagents affords α-amino esters. M2AA can form thermosensitive copolymers with methyl acrylate.
Methyl ester of 2-acetamidoacrylate . methyl 2-acetamidoacrylate (Me-2-AA) is a di-unsaturated α-amino acid derivative. methyl-2-acetamidoacrylate exihibits anti -inflammatory properties, it is very effective against lipopolysaccharide (LPS)- induced nitric oxide production by RAW 264.
應用
Methyl 2-acetamidoacrylate can undergo [2+2] cycloaddition (Michael–Dieckmann-type reaction) with ketene diethyl acetal to yield the cyclobutane core. It may be used in rhodium-catalyzed 2-alkenylpyrrole formation.
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
235.4 °F - closed cup
閃點(°C)
113 °C - closed cup
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
Thermosensitive properties of a novel poly(methyl 2-acetamidoacrylate-co-methyl acrylate)
Okamura H, et al.
European Polymer Journal, 38(4), 639-644 (2002)
Madeleine E Kieffer et al.
Journal of the American Chemical Society, 134(11), 5131-5137 (2012-03-07)
The tandem Friedel-Crafts conjugate addition/asymmetric protonation reaction between 2-substituted indoles and methyl 2-acetamidoacrylate is reported. The reaction is catalyzed by (R)-3,3'-dibromo-BINOL in the presence of stoichiometric SnCl(4), and is the first example of a tandem conjugate addition/asymmetric protonation reaction using
Joel A Tang et al.
Chemical communications (Cambridge, England), 47(3), 958-960 (2010-11-17)
A system that provides a sustained hyperpolarized (1)H NMR signal in an aqueous medium is reported. The enhanced signal lasts much longer than typical (1)H T(1) values, uncovering new possibilities for implementing hyperpolarized (1)H NMR/MRI experiments or performing kinetics studies
Synthesis of ?-amino acid derivatives by copper(I)-catalyzed conjugate addition of grignard reagents to methyl 2-acetamidoacrylate,
Cardellicchio C, et al.
Tetrahedron, 26(36), 4387-4390 (1985)
The Vinyl Moiety as a Handle for Regiocontrol in the Preparation of Unsymmetrical 2, 3?Aliphatic?Substituted Indoles and Pyrroles.
Huestis MP, et al.
Angewandte Chemie (International Edition in English), 123(6), 1374-1377 (2011)
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门