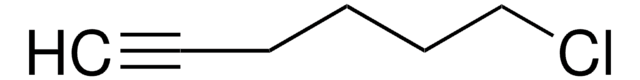
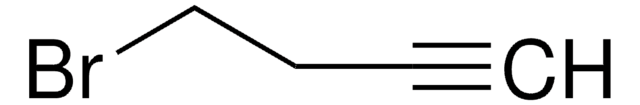
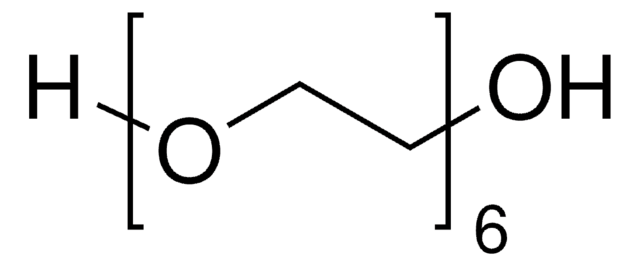
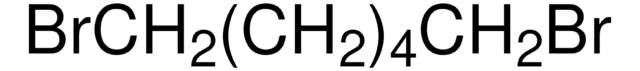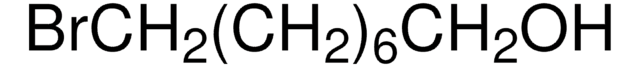推荐产品
品質等級
化驗
97%
形狀
liquid
折射率
n20/D 1.467 (lit.)
密度
1.139 g/mL at 25 °C (lit.)
官能基
alkyl halide
allyl
bromo
SMILES 字串
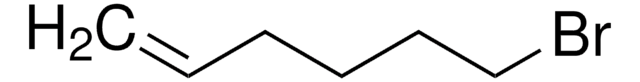
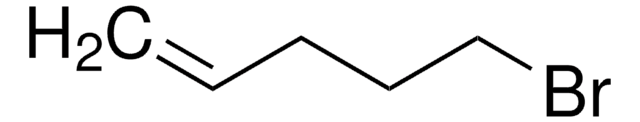
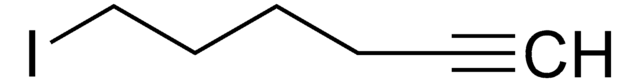
BrCCCCCCC=C
InChI
1S/C8H15Br/c1-2-3-4-5-6-7-8-9/h2H,1,3-8H2
InChI 密鑰
SNMOMUYLFLGQQS-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
應用
8-Bromo-1-octene has been used in preparation of polymerizable ligand, required for the synthesis of quantum dot-labelled polymer beads. Grignard reagent derived from 8-bromo-1-octene has been used in the synthesis of (2S,3S,5R)-5-[(1R)-1-hydroxy-9-decenyl]-2-pentyltetrahydro-3-furanol. It has been used as building block in natural product synthesis.
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
172.4 °F - closed cup
閃點(°C)
78 °C - closed cup
個人防護裝備
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
其他客户在看
Enantiocontrolled synthesis of C-19 tetrahydrofurans isolated from the marine alga Notheia anomala.
Garci'a C, et al.
Tetrahedron Letters, 41(21), 4127-4130 (2000)
Christophe Dubost et al.
Organic letters, 8(22), 5137-5140 (2006-10-20)
The total synthesis of the polyhydroxylated macrolide (+)-aspicilin 5 is described using as a key step a highly diastereoselective allylation of aldehyde 6 with the uniquely functionalized allylstannane 1. (+)-Aspicilin is obtained in 18 steps and 10% overall yield. [structure:
Carolyn A Leverett et al.
The Journal of organic chemistry, 71(22), 8591-8601 (2006-10-27)
cis-2-Methyl-6-substituted piperidin-3-ol alkaloids of the Cassia and Prosopis species are readily prepared by a combination of an aza-Achmatowicz oxidative rearrangement and dihydropyridone reduction followed by a stereoselective allylsilane addition to a N-sulfonyliminium ion. The stereochemical outcome of the reduction reaction
Paul O'Brien et al.
Chemical communications (Cambridge, England), (20)(20), 2532-2533 (2003-11-05)
CdSe quantum dots with polymerisable ligands have been incorporated into polystyrene beads, via a suspension polymerisation reaction, as a first step towards the optical encoding of solid supports for application in solid phase organic chemistry.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门