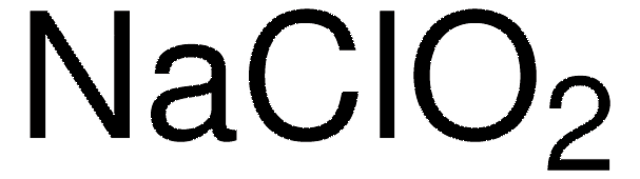8.14815
Sodium chlorite
(25% solution in water) for synthesis
Synonym(s):
Chlorous acid, sodium salt
About This Item
Recommended Products
vapor pressure
20 hPa ( 20 °C)
Quality Level
form
liquid
potency
1136 mg/kg LD50, oral (Rat)
pH
12-13 (20 °C in H2O)
bp
≥100 °C/1013 hPa
mp
<-3 °C
density
1.20-1.25 g/cm3 at 20 °C
storage temp.
2-30°C
Application
- Aldehydes to corresponding carboxylic acids under mild reaction conditions in the presence of scavengers such as sulfamic acid or resorcinol.
- Primary alcohols to carboxylic acids in the presence of 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) and NaOCl.
- Aliphatic and aromatic thiols to the corresponding disulfides.
- Aryl boronic acids to phenols via ipso-hydroxylation.
- Cyclic benzylic ethers and N-protected cyclic benzylic amines to benzolactones and benzolactams.
- Alkenes to epoxides using aldehydes as promoters.
- Alkenes and cycloalkenes to 1, 2 diols via osmium-catalyzed Sharpless asymmetric dihydroxylation.
It can also be used in combination with Br2 to synthesize 2-carboxyazetidin-2-ones from furanylazetidinones.
Analysis Note
Density (d 20 °C/ 4 °C): 1.200 - 1.220
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 4 Oral - Aquatic Acute 1 - Eye Dam. 1 - Met. Corr. 1 - Skin Corr. 1 - STOT RE 2
Target Organs
spleen
Supplementary Hazards
Storage Class Code
6.1D - Non-combustible, acute toxic Cat.3 / toxic hazardous materials or hazardous materials causing chronic effects
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service