640441
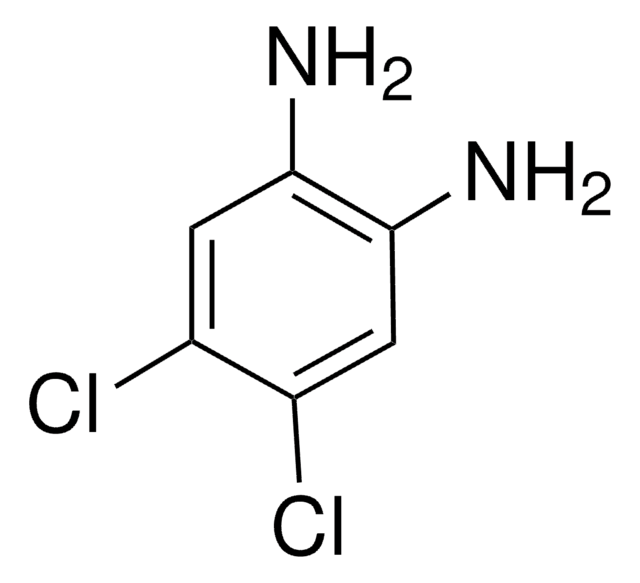
4-Bromo-1,2-diaminobenzene
97%
Synonym(s):
4-Bromo-2-aminoaniline, 4-Bromo-o-phenylenediamine
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
C6H7BrN2
CAS Number:
Molecular Weight:
187.04
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
solid
mp
65-69 °C (dec.) (lit.)
functional group
bromo
SMILES string
Nc1ccc(Br)cc1N
InChI
1S/C6H7BrN2/c7-4-1-2-5(8)6(9)3-4/h1-3H,8-9H2
InChI key
WIHHVKUARKTSBU-UHFFFAOYSA-N
Related Categories
General description
4-Bromo-1,2-diaminobenzene can be obtained from 1,2-diaminobenzene via acetylation followed by bromination and alkaline hydrolysis.
4-Bromo-1,2-diaminobenzene is used as a precursor in the production of tetra-dentate unsymmetrical Schiff base.
4-Bromo-1,2-diaminobenzene is used as a precursor in the production of tetra-dentate unsymmetrical Schiff base.
Application
4-Bromo-1,2-diaminobenzene can be used as a precursor for preparing fluorescent dipolar quinoxaline derivatives, which can find applications as potential emissive and electron-transport materials. It can also be used in the synthesis of 6-bromo-2-methylbenzimidazole.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Oral - Eye Irrit. 2 - Skin Irrit. 2 - Skin Sens. 1
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
"Guanidine chemistry"
Ishikawa T, et al.
Chemical & Pharmaceutical Bulletin, 58(12), 1555-1564 (2010)
"The synthesis and characterization of novel dipolar fluorescent materials based on a quinoxaline core"
Wang H, et al.
Dyes and Pigments, 83(03), 269-275 (2009)
"A PdII Complex Bearing a Benzimidazole-Derived Ligand with Potentially ?Mesoionic and Remote? Character and Its Catalytic Activity"
Huynh VH, et al.
European Journal of Organic Chemistry, 2013(26), 4654-4661 (2013)
Erin Wachter et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 22(2), 550-559 (2015-11-13)
Recognition and regulation of G-quadruplex nucleic acid structures is an important goal for the development of chemical tools and medicinal agents. The addition of a bromo-substituent to the dipyridylphenazine (dppz) ligands in the photophysical "light switch", [Ru(bpy)2 dppz](2+) , and
Jacek E Nycz et al.
Molecules (Basel, Switzerland), 24(22) (2019-11-27)
New approaches to the synthesis of 4,7-dichloro-1,10-phenanthrolines and their corresponding 9H-carbazol-9-yl-, 10H-phenothiazin-10-yl- and pyrrolidin-1-yl derivatives were developed. Their properties have been characterized by a combination of several techniques: MS, HRMS, GC-MS, electronic absorption spectroscopy and multinuclear NMR in both solution
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service