250279
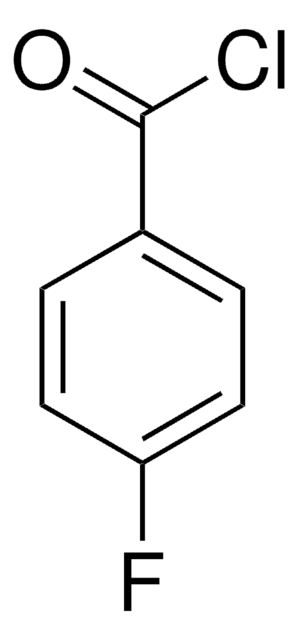
3-(Trifluoromethyl)benzoyl chloride
98%
Synonym(s):
α,α,α-Trifluoro-m-toluoyl chloride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CF3C6H4COCl
CAS Number:
Molecular Weight:
208.56
Beilstein:
391266
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
liquid
refractive index
n20/D 1.477 (lit.)
bp
184-186 °C/750 mmHg (lit.)
density
1.383 g/mL at 25 °C (lit.)
functional group
acyl chloride
fluoro
SMILES string
FC(F)(F)c1cccc(c1)C(Cl)=O
InChI
1S/C8H4ClF3O/c9-7(13)5-2-1-3-6(4-5)8(10,11)12/h1-4H
InChI key
RUJYJCANMOTJMO-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
3-(Trifluoromethyl)benzoyl chloride has been used in the preparation of intermediates, required for synthesis of C-2 and C-3 substituted pyrazolo[1,5-a]pyrimidines.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Dan M Berger et al.
Bioorganic & medicinal chemistry letters, 19(23), 6519-6523 (2009-10-30)
As part of our research effort to discover B-Raf kinase inhibitors, we prepared a series of C-3 substituted N-(3-(pyrazolo[1,5-a]pyrimidin-7-yl)phenyl)-3-(trifluoromethyl)benzamides. X-ray crystallography studies revealed that one of the more potent inhibitors (10n) bound to B-Raf kinase without forming a hinge-binding hydrogen
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service