EMS0006
Recombinant Trypsin
Proteomics Grade, lyophilized powder, recombinant, expressed in Pichia pastoris
Synonym(s):
Mass Spectrometry Trypsin, Proteomics grade Trypsin, rTrypsin
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
UNSPSC Code:
12352204
NACRES:
NA.26
Recommended Products
Related Categories
General description
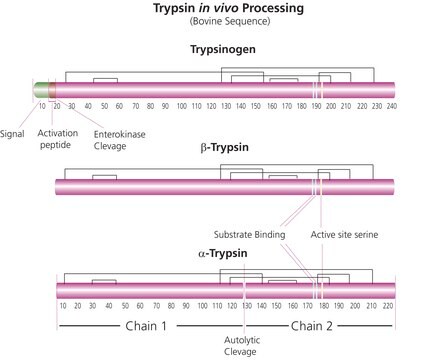
Trypsin is a major proteolytic enzyme, synthesized as a preproenzyme by pancreas and is stored as proenzyme trypsinogen in secretory granules. Trypsin belongs to the family of serine proteases that are characterized by the catalytic triad His57, Asp102 and Ser195. Trypsin is routinely used in proteomics research for peptide mapping and protein sequence work due to its highly specific cleavage resulting in a limited number of tryptic peptides. Trypsin is a pancreatic serine endoprotease which hydrolyzes peptide bonds specifically at the carboxyl side of arginine and lysine residues. The rate of hydrolysis is slower if an acidic residue is on either side of the cleavage site and cleavage may not occur if a proline residue is on the carboxyl side. The enzyme also exhibits esterase and amidase activities. Trypsin has an average molecular mass of 23.29 kDa and a pH optimum near 8.0. This product is prepared from recombinant trypsin, porcine sequence. It is naturally devoid of chymotryptic activity. This high-quality trypsin is suitable for proteomics use.
Biochem/physiol Actions
Trypsin plays an important role in the digestion of consumed protein and contributes to the activation of other proteolytic enzymes like chemotrypsin and elastase.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 2 - Eye Dam. 1 - Met. Corr. 1 - Resp. Sens. 1 - Skin Corr. 1A - Skin Sens. 1 - STOT SE 3
Target Organs
Respiratory system
Supplementary Hazards
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Structural properties of trypsin from cold-adapted fish, arabesque greenling (Pleurogrammus azonus)
Kanno G, et al.
European Food Research and Technology, 232(3), 381-388 (2011)
Molecular characterization and gene expression of six trypsinogens in the flatfish Senegalese sole (Solea senegalensis Kaup) during larval development and in tissues
Manchado M, et al.
Comparative Biochemistry and Physiology. Part B, Biochemistry & Molecular Biology, 149(2), 334-344 (2008)
Handbook of Proteolytic Enzymes (2012)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service