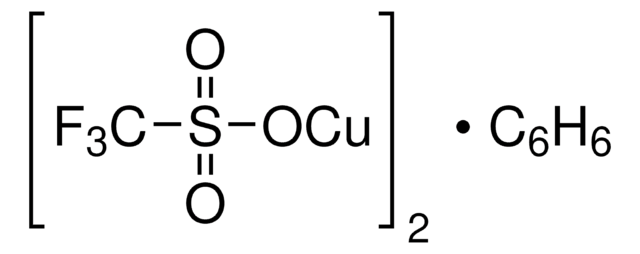283673
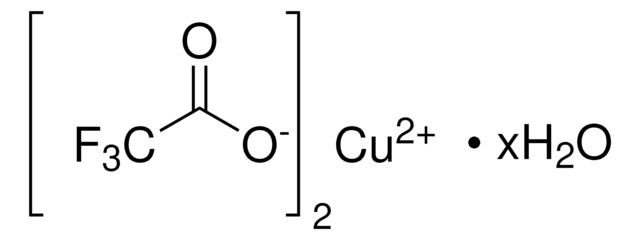
Copper(II) trifluoromethanesulfonate
98%
Synonym(s):
Cu(OTf)2, Copper(II) triflate, Cupric trifluoromethanesulfonate, Trifluoromethanesulfonic acid copper(II) salt
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
(CF3SO3)2Cu
CAS Number:
Molecular Weight:
361.68
Beilstein:
4028198
EC Number:
MDL number:
UNSPSC Code:
12161600
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
reaction suitability
core: copper
reagent type: catalyst
mp
≥300 °C
SMILES string
FC(F)(F)S(=O)(=O)O[Cu]OS(=O)(=O)C(F)(F)F
InChI
1S/2CHF3O3S.Cu/c2*2-1(3,4)8(5,6)7;/h2*(H,5,6,7);/q;;+2/p-2
InChI key
SBTSVTLGWRLWOD-UHFFFAOYSA-L
Looking for similar products? Visit Product Comparison Guide
Application
Catalyst for:
- Mannich condensation
- Annulative amination
- Friedel-Crafts reaction
- Henry reaction
- Hypervalent iodine reagent-mediated preparation of carbazoles
- Intramolecular oxidative C-N bond formation for the synthesis of carbazoles
Catalyzes the efficient addition of trimethylsilyl cyanide to carbonyl compounds.
related product
Product No.
Description
Pricing
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Ahmed M Senan et al.
Journal of agricultural and food chemistry, 65(32), 6912-6918 (2017-07-19)
Utilization of renewable biomass to partly replace the fossil resources in industrial applications has attracted attention due to the limited fossil feedstock with the increased environmental concerns. This work introduced a modified Wacker-type oxidation for transformation of unsaturated fatty acids/esters
Saravanan, P. et al.
Tetrahedron Letters, 39, 3823-3823 (1998)
Dario Formenti et al.
Chemical reviews, 119(4), 2611-2680 (2018-12-06)
The reduction of nitro compounds to the corresponding amines is one of the most utilized catalytic processes in the fine and bulk chemical industry. The latest development of catalysts with cheap metals like Fe, Co, Ni, and Cu has led
Nicole W G Smits et al.
Inorganic chemistry, 58(19), 13007-13019 (2019-09-25)
The structure of the copper complex of the 6-((1-butanethiol)oxy)-tris(2-pyridylmethyl)amine ligand (Cu-tmpa-O(CH2)4SH) anchored to a gold surface has been investigated. To enable covalent attachment of the complex to the gold surface, a heteromolecular self-assembled monolayer (SAM) of butanethiol and a thiol-substituted
Juan Ramírez et al.
Inorganic chemistry, 48(6), 2456-2463 (2009-03-10)
A novel class of ditopic ligands, 1, was synthesized by the reaction of 2,5-pyrazine-dicarboxaldehyde with 2 equiv of acyl-/aroyl-hydrazine. Their structures were confirmed by 1D and 2D NMR and by X-ray crystallography. They gave heteroleptic Cu(II) dinuclear rack-like complexes of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service