233587
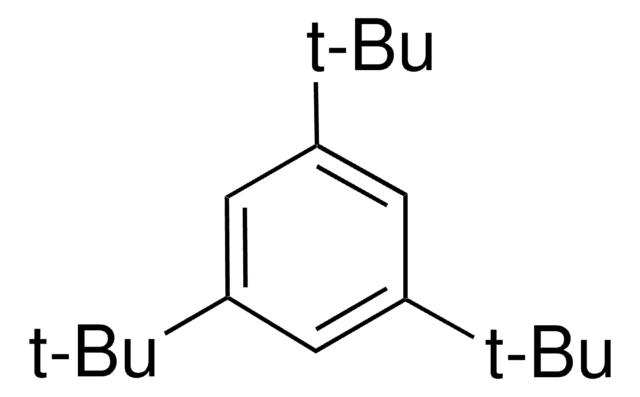
1-tert-Butyl-3,5-dimethylbenzene
98%
Synonym(s):
5-tert-Butyl-m-xylene, sym.-tert-Butyl-m-xylene
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
(CH3)3CC6H3(CH3)2
CAS Number:
Molecular Weight:
162.27
Beilstein:
1853314
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
liquid
refractive index
n20/D 1.495 (lit.)
bp
205-206 °C (lit.)
density
0.867 g/mL at 25 °C (lit.)
SMILES string
Cc1cc(C)cc(c1)C(C)(C)C
InChI
1S/C12H18/c1-9-6-10(2)8-11(7-9)12(3,4)5/h6-8H,1-5H3
InChI key
FZSPYHREEHYLCB-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
1-tert-Butyl-3,5-dimethylbenzene participates in the cascade diarylation of N-phenylacetamides with non-prefunctionalized arenes.
Application
1-tert-Butyl-3,5-dimethylbenzene has been used in the synthesis of bulky, multi-alkylated diaryl disulfides such as bis(4-tert-butyl-2,6-dimethylphenyl) disulfide and bis(2,4,6-triisopropylphenyl) disulfide.
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
154.4 °F - closed cup
Flash Point(C)
68 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Rajarshi Samanta et al.
Chemical communications (Cambridge, England), 48(26), 3194-3196 (2012-02-15)
A new atom-economical process of direct oxidative intermolecular functionalization of aniline derivatives by simple arenes was developed. The products were formed in a highly regioselective manner under metal-free conditions at ambient temperature.
Teruo Umemoto et al.
Journal of the American Chemical Society, 132(51), 18199-18205 (2010-12-04)
Versatile, safe, shelf-stable, and easy-to-handle fluorinating agents are strongly desired in both academic and industrial arenas, since fluorinated compounds have attracted considerable interest in many areas, such as drug discovery, due to the unique effects of fluorine atoms when incorporated
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service