123617
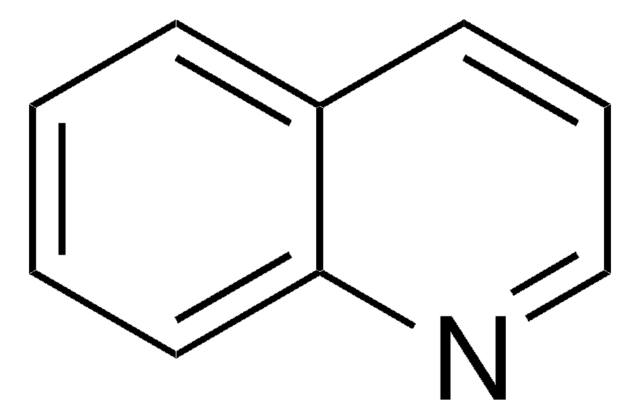
Benzo[h]quinoline
97%
Synonym(s):
1-Naphthoquinoline, 7,8-Benzoquinoline
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C13H9N
CAS Number:
Molecular Weight:
179.22
Beilstein:
120249
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
solid
bp
338 °C/719 mmHg (lit.)
mp
48-50 °C (lit.)
SMILES string
c1ccc2c(c1)ccc3cccnc23
InChI
1S/C13H9N/c1-2-6-12-10(4-1)7-8-11-5-3-9-14-13(11)12/h1-9H
InChI key
WZJYKHNJTSNBHV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
Benzo[h]quinoline was used to study the mutagenic activities of benzo[f]quinoline, benzo[h]quinolone and a number of their derivatives in strain TA 100 of Salmonella typhimurium. It was used in determination of nitrogen-containing polynuclear aromatic hydrocarbons in the gaseous products of the thermal degradation of polymers by HPLC- fluorescence detection. It was used as starting reagent for the synthesis of osmium and ruthenium complexes containing an N-heterocyclic carbene ligand.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
E J LaVoie et al.
Carcinogenesis, 4(9), 1133-1138 (1983-09-01)
Benzo[f]quinoline and benzo[h]quinoline are widespread environmental pollutants which have been found to be mutagenic. The metabolism of benzo[f]quinoline and benzo[h]quinoline was investigated using a liver homogenate from Aroclor-pretreated rats. The metabolites of benzo[f]quinoline which were identified were 7,8-dihydroxy-7,8-dihydrobenzo[f]quinoline, 9,10-dihydroxy-9,10-dihydrobenzo[f]quinoline, 7-hydroxybenzo[f]quinoline
Marko Weimar et al.
Organic & biomolecular chemistry, 11(1), 31-34 (2012-10-17)
The challenging coupling of 10-halobenzo[h]quinolines with ortho-substituted aryl boronic acids has been achieved using Pd(OAc)(2)/P(O)Ph(3) as the catalytic system. High yields were obtained for diversely functionalised substrates under mild reaction conditions.
E J LaVoie et al.
Japanese journal of cancer research : Gann, 78(2), 139-143 (1987-02-01)
The environmental occurrence and mutagenic activity of quinoline and benzoquinolines are well-documented. In this study, the relative carcinogenic activities of quinoline, benzo[f]quinoline, benzo[h]quinoline, and phenanthridine were evaluated in newborn mice. Mice were injected intraperitoneally on the first, eighth, and fifteenth
Hanumantharao Paritala et al.
Bioorganic & medicinal chemistry letters, 19(6), 1584-1587 (2009-02-27)
G-quadruplexes are unusual structures formed from guanine-rich sequences of nucleic acids. G-quadruplexes have been postulated to play important roles in a number of biological systems including gene regulation and the inhibition of enzyme function. Recently, our laboratory reported on the
John B Sutherland et al.
Applied microbiology and biotechnology, 67(3), 405-411 (2005-04-28)
Cultures of Umbelopsis ramanniana (=Mucor ramannianus) were grown in fluid Sabouraud medium for 3 days, dosed with 0.23 mM benzo[f]quinoline, benzo[h]quinoline, or phenanthridine (benzo[c]quinoline), and incubated for another 18 days. Cultures were extracted and metabolites (66-75% of the UV absorbance)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service


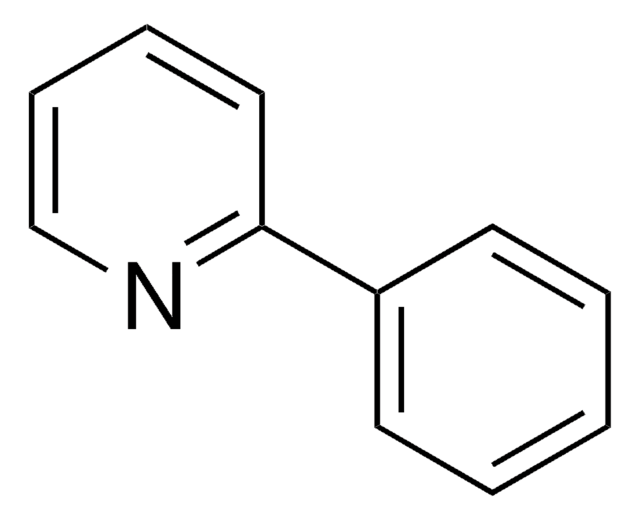

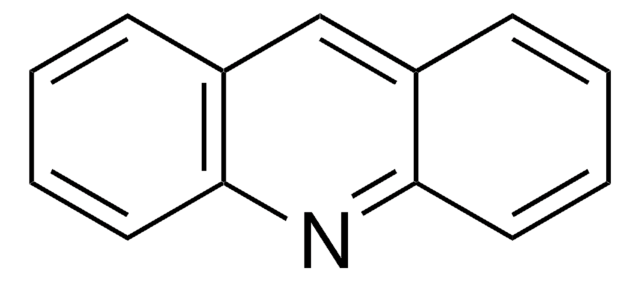
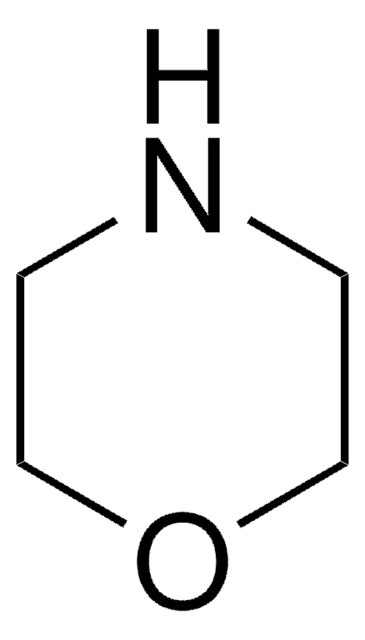
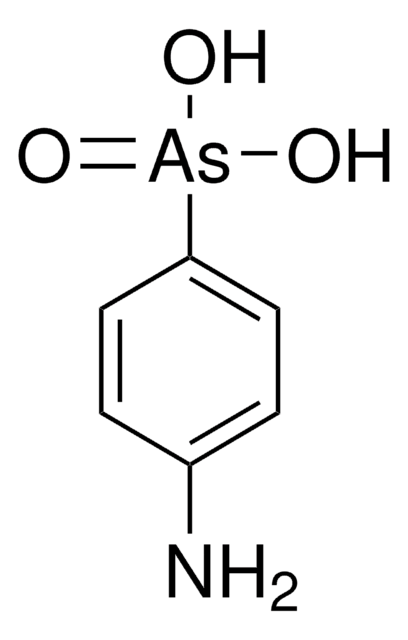
![Dibenz[c,h]acridine BCR®, certified reference material](/deepweb/assets/sigmaaldrich/product/structures/364/643/698df9fb-5b7d-467a-b47e-c8318e2ed298/640/698df9fb-5b7d-467a-b47e-c8318e2ed298.png)
![Benz[g]isoquinoline-5,10-dione 99%](/deepweb/assets/sigmaaldrich/product/structures/484/029/288c4a9d-19c2-4b51-82c1-f43b50ea05b0/640/288c4a9d-19c2-4b51-82c1-f43b50ea05b0.png)