All Photos(1)
About This Item
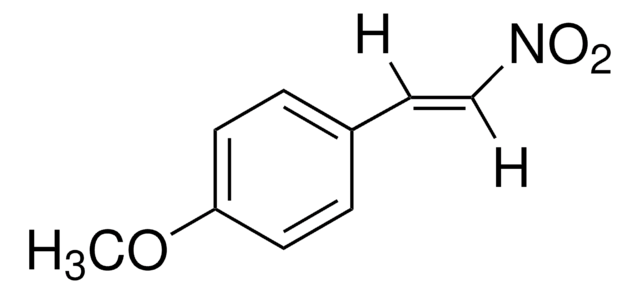
Linear Formula:
C6H5CH=C(CH3)NO2
CAS Number:
Molecular Weight:
163.17
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
form
solid
mp
63-65 °C (lit.)
storage temp.
2-8°C
SMILES string
[H]\C(=C(\C)[N+]([O-])=O)c1ccccc1
InChI
1S/C9H9NO2/c1-8(10(11)12)7-9-5-3-2-4-6-9/h2-7H,1H3/b8-7+
InChI key
WGSVFWFSJDAYBM-BQYQJAHWSA-N
General description
trans-β-Methyl-β-nitrostyrene (1-phenyl-2-nitropropene), a nitrostyrene derivative is an α,β-disubstituted nitroalkene. It has been synthesized by reacting benzaldehyde with nitroethane and butylamine. Spectroscopic analysis of 1-phenyl-2-nitropropene has been done using FT-IR, FT-Raman, NMR and UV.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
A Mori et al.
Chemical & pharmaceutical bulletin, 38(12), 3449-3451 (1990-12-01)
Microbial reduction of 1-phenyl-2-nitro-1-propene (3) was carried out using 57 strains of yeast, 40 strains of aerobic and facultatively anaerobic bacteria and 40 strains of strictly anaerobic bacteria. Nine strains of yeast (Candida tropicalis, etc.,) had the ability to reduce
Gas chromatographic and mass spectral analysis of amphetamine products synthesized from 1-phenyl-2-nitropropene.
DeRuiter J, et al.
Journal of Chromatographic Science, 32(11), 511-519 (1994)
Diego Romano et al.
Microbial cell factories, 13, 60-60 (2014-04-29)
Old Yellow Enzymes (OYEs) are flavin-dependent enoate reductases (EC 1.6.99.1) that catalyze the stereoselective hydrogenation of electron-poor alkenes. Their ability to generate up to two stereocenters by the trans-hydrogenation of the C = C double bond is highly demanded in asymmetric synthesis.
Organocatalytic diastereo- and enantioselective sulfa-Michael addition to α,β-disubstituted nitroalkenes
Pei QL, et al.
Tetrahedron, 69, 5367-5373 (2013)
Tundo P and Andraos J
Green Syntheses, 119-119 (2014)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service