206547
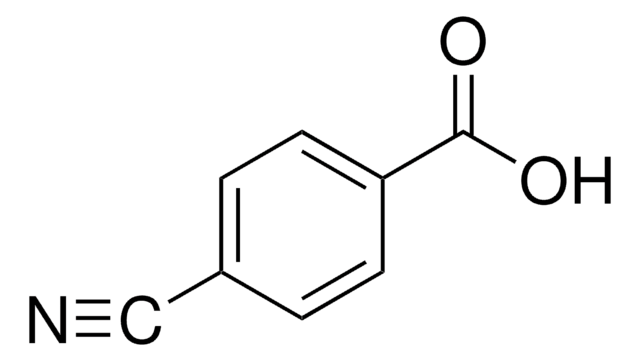
4-Iodobenzoic acid
98%
Synonym(s):
4-Iodobenzoic acid, Iodobenzene-4-carboxylic acid, p-Iodobenzenecarboxylic acid, p-Iodobenzoic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
IC6H4CO2H
CAS Number:
Molecular Weight:
248.02
Beilstein:
1860232
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
mp
270-273 °C (lit.)
functional group
carboxylic acid
SMILES string
OC(=O)c1ccc(I)cc1
InChI
1S/C7H5IO2/c8-6-3-1-5(2-4-6)7(9)10/h1-4H,(H,9,10)
InChI key
GHICCUXQJBDNRN-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
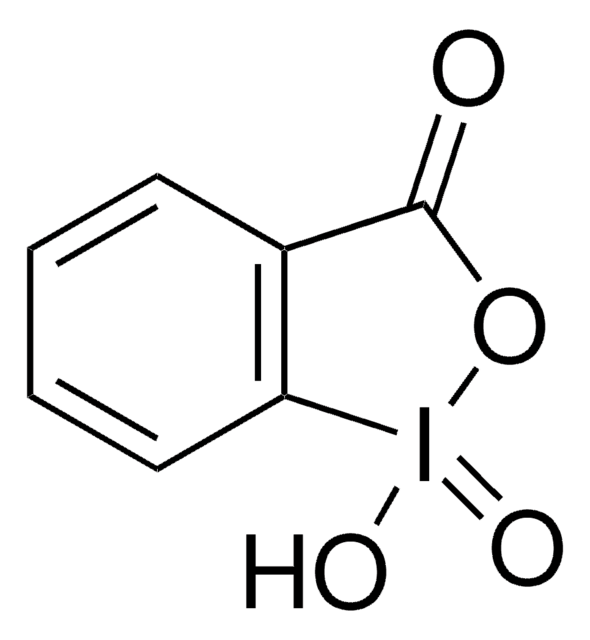
4-Iodobenzoic acid was used in the synthesis of [hydroxy(4-carboxyphenyl)iodonium]ion in situ that helps in the cleavage of a variety of alkenes.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Prem P Thottumkara et al.
Organic letters, 12(24), 5640-5643 (2010-11-18)
A facile and operationally convenient catalytic procedure for oxidative cleavage of alkenes is described. In situ formed [hydroxy(4-carboxyphenyl)iodonium]ion, 2, from the oxidation of 4-iodobenzoic acid, 1, has been shown to facilitate the cleavage of a variety of alkenes in presence
Peggy E Williams et al.
Journal of the American Society for Mass Spectrometry, 29(9), 1848-1860 (2018-06-06)
In the gas phase, arylperoxyl forming reactions play a significant role in low-temperature combustion and atmospheric processing of volatile organic compounds. We have previously demonstrated the application of charge-tagged phenyl radicals to explore the outcomes of these reactions using ion
Toshiaki Taira et al.
Journal of oleo science, 67(9), 1107-1115 (2018-09-04)
In this study, an N-heterocyclic carbene (NHC)-based metallosurfactant (MS), NHC-PdMS, was synthesized, where Pd(II) was bound to the NHC framework via a robust Pd-carbene bond with NEt3 as a co-ligand. Surface tension measurements revealed that the critical micelle concentration (CMC)
Zengyan Wei et al.
Nature communications, 5, 3870-3870 (2014-05-16)
The shape-controlled synthesis of nanoparticles was established in single-phase solutions by controlling growth directions of crystalline facets on seed nanocrystals kinetically; however, it was difficult to rationally predict and design nanoparticle shapes. Here we introduce a methodology to fabricate nanoparticles
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service