All Photos(1)
About This Item
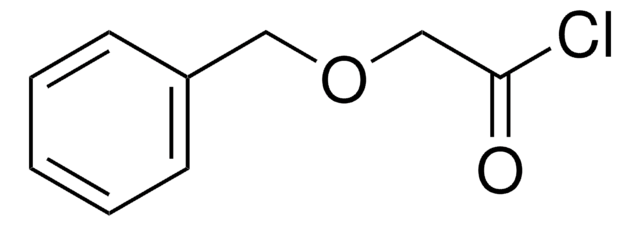
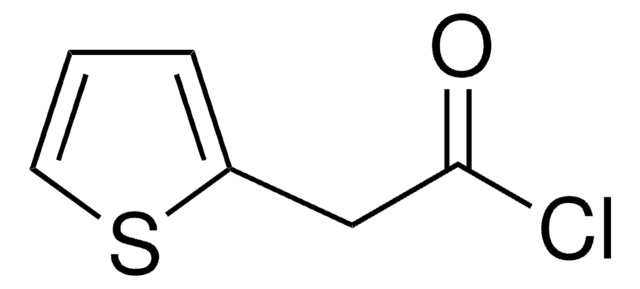
Linear Formula:
C6H5OCH2COCl
CAS Number:
Molecular Weight:
170.59
Beilstein:
607585
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
refractive index
n20/D 1.534 (lit.)
bp
225-226 °C (lit.)
density
1.235 g/mL at 25 °C (lit.)
functional group
acyl chloride
SMILES string
ClC(=O)COc1ccccc1
InChI
1S/C8H7ClO2/c9-8(10)6-11-7-4-2-1-3-5-7/h1-5H,6H2
InChI key
PKUPAJQAJXVUEK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Phenoxyacetyl chloride was used in the synthesis of:
- series of macrocyclic bis-β-lactams
- 5-phenyl-6-epiphenoxymethylpenicillin benzyl ester
- N-protected guanosine derivatives, useful in RNA synthesis
- phenyloxyketene, for cycloaddition to imines leading to β-lactams
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Corr. 1B - STOT SE 3
Target Organs
Respiratory system
Supplementary Hazards
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Natarajan Arumugam et al.
Medicinal chemistry (Shariqah (United Arab Emirates)), 10(7), 730-737 (2014-02-27)
A series of macrocyclic bis-β-lactams has been synthesized in three good yielding steps using a Staudinger [2+2] cycloaddition reaction of ketene derived from phenoxyacetyl chloride as the key step. The reaction provided a diastereomeric mixture of cis-anti-cis (C2-symmetry) and cis-syn-cis
Tetrahedron, 63, 3380-3380 (2007)
H Vanderhaeghe et al.
Journal of medicinal chemistry, 18(5), 486-490 (1975-05-01)
Cycloaddition of azidoacetyl chloride to benzyl D-5,5-dimethyl-5-phenyl-2-thiazoline-4-carboxylate (1a) gave 5-phenyl-6alpha-azidopenicillanate (2a). By catalytic reduction of 2a and reaction with phenoxyacetyl chloride, 5-phenyl-6-epiphenoxymethylpenicillin benzyl ester (4a) was obtained. Oxidation of 4a gave the sulfoxide 6, which was isomerized in the presence
Yupeng Fan et al.
Organic letters, 6(15), 2555-2557 (2004-07-17)
[reaction: see text] The formation of a guanosine derivative silylated at both the O6 and amino groups was identified by (15)N NMR. This intermediate allows facile reaction with acetyl chloride or phenoxyacetyl chloride to give in high yield the corresponding
Tetrahedron Letters, 48, 1657-1657 (2007)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service