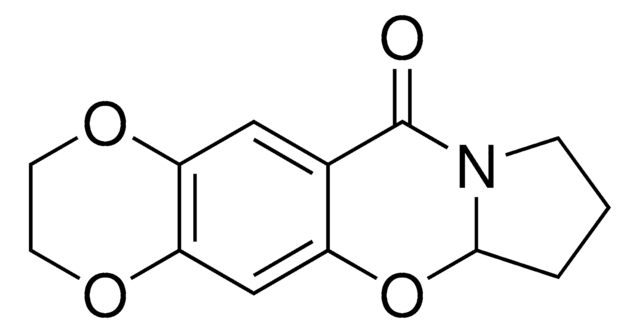
C271
CX546
≥98% (HPLC), solid
Synonym(s):
1-(1,4-Benzodioxan-6-ylcarbonyl)piperidine
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C14H17NO3
CAS Number:
Molecular Weight:
247.29
MDL number:
UNSPSC Code:
12352200
PubChem Substance ID:
NACRES:
NA.77
Recommended Products
Quality Level
Assay
≥98% (HPLC)
form
solid
storage condition
protect from light
color
white to off-white
solubility
DMSO: ≥10 mg/mL
storage temp.
2-8°C
SMILES string
O=C(N1CCCCC1)c2ccc3OCCOc3c2
InChI
1S/C14H17NO3/c16-14(15-6-2-1-3-7-15)11-4-5-12-13(10-11)18-9-8-17-12/h4-5,10H,1-3,6-9H2
InChI key
LJUNPHMOGNFFOS-UHFFFAOYSA-N
Gene Information
human ... GRIA1(2890) , GRIA2(2891) , GRIA3(2892) , GRIA4(2893)
General description
CX546, a benzoylpiperidine derivative ampakine is an analog of CX516.
Application
CX546 has been used as positive allosteric modulator for the glutamatergic receptor α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) in mice.
Biochem/physiol Actions
CX546 has antipsychotic functionality and has the potential to treat schizophrenia. It improves the defects associated with the prepulse inhibition (PPI) and latent inhibition (LI) in mice lacking metabotropic glutamate receptor type 5 (mGluR5). Additionally, CX546 potentiates synaptic plasticity, elicits neuroprotection and promotes the neurotrophin expression.
Positive AMPA glutamate receptor modulator.
Caution
Photosensitive
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Amy C Arai et al.
The Journal of pharmacology and experimental therapeutics, 303(3), 1075-1085 (2002-11-20)
CX516 (BDP-12) and CX546, two first-generation benzamide-type AMPA receptor modulators, were compared with regard to their influence on AMPA receptor-mediated currents, autaptic responses in cultured hippocampal neurons, hippocampal excitatory postsynaptic currents, synaptic field potentials, and agonist binding. The two drugs
Laureen D Hachem et al.
Stem cells and development, 26(23), 1675-1681 (2017-09-28)
Transplantation of neural stem/progenitor cells (NSPCs) following spinal cord injury (SCI) is a promising strategy to enhance regeneration but is limited by poor survival of grafted cells. Determining methods to enhance survival of NSPCs is therefore essential. Positive modulation of
C Lu et al.
Molecular psychiatry, 21(2), 159-168 (2015-11-26)
Numerous risk genes have recently been implicated in susceptibility to autism and schizophrenia. Translating such genetic findings into disease-relevant neurobiological mechanisms is challenging due to the lack of throughput assays that can be used to assess their functions on an
Ampakines: Labelling with 11C for PET distribution studies.
Rogers, et al.
Journal of Labelled Compounds & Radiopharmaceuticals, 40, 645-647 (1997)
Chen Su et al.
Anesthesiology, 125(5), 1030-1043 (2016-10-19)
AMPAkines augment the function of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors in the brain to increase excitatory outputs. These drugs are known to relieve persistent pain. However, their role in acute pain is unknown. Furthermore, a specific molecular and anatomic target for
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service