General description
Lot specific orders are not able to be placed through the web. Contact your local sales rep for more details.
Cell Line Origin
Umbilical Cord
Application
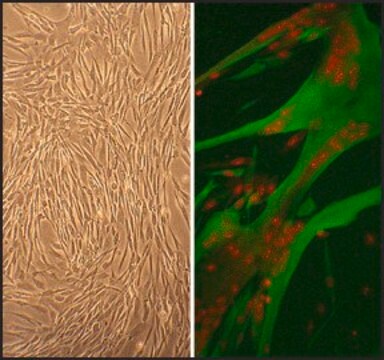
Mesenchymal Stem Cells (MSC), also termed Mesenchymal Stromal Cells, are multipotent cells that can differentiate into a variety of cell types and have the capacity for self renewal. MSC have been shown to differentiate in vitro or in vivo into adipocytes, chondrocytes, osteoblasts, myocytes, neurons, hepatocytes, and pancreatic islet cells. Optimized PromoCell media are available to support both the growth of MSC and their differentiation into several different lineages. Recent experiments suggest that differentiation capabilities into diverse cell types vary between MSC of different origin.Human Mesenchymal Stem Cells (hMSC-UC) are harvested from normal human umbilical cord matrix (Wharton′s jelly) of individual donors and are provided in a proliferating format. The cells are tested for their ability to differentiate in vitro into adipocytes, chondrocytes and osteoblasts. The cells show a verified marker expression profile that complies with ISCT recommendations, providing well characterized cells (Cytotherapy (2006) Vol. 8, No. 4, 315-317).
Quality
Rigid quality control tests are performed for each lot of Mesenchymal Stem Cells. They are tested for cell morphology, proliferation potential, adherence rate, and viability. Furthermore, they are characterized by flow cytometric analysis of a comprehensive panel of markers, namely CD73/CD90/CD105 and CD14/CD19/CD34/CD45/HLA-DR as proposed by the ISCT. Differentiation assays into adipogenic, osteogenic and chrondrogenic are performed for each lot under culture conditions without antibiotics and antimycotics. In addition, all cells have been tested for the absence of HIV-1, HIV-2, HBV, HCV, HTLV-1 and HTLV-2, and microbial contaminants (fungi, bacteria, and mycoplasma).
Warning
Although tested negative for HIV-1, HIV-2, HBV, HCV, HTLV-1 and HTLV-2, the cells – like all products of human origin – should be handled as potentially infectious. No test procedure can completely guarantee the absence of infectious agents.
Subculture Routine
Click
here for more information.
Other Notes
Recommended Plating Density: 4000 cells per cm2Tested Markers: CD105 positive, CD73 positive, CD90 positive, CD45 negative, CD34 negative, CD14 negative, CD19 negative, HLA-DR negativeDifferentiation capacity to adipogenic, osteogenic and chondrogenic cells
Recommended products
Recommended Primary Cell Culture Media:
LinkRecommended Primary Cell Culture Media:
LinkDisclaimer
RESEARCH USE ONLY. This product is regulated in France when intended to be used for scientific purposes, including for import and export activities (Article L 1211-1 paragraph 2 of the Public Health Code). The purchaser (i.e. enduser) is required to obtain an import authorization from the France Ministry of Research referred in the Article L1245-5-1 II. of Public Health Code. By ordering this product, you are confirming that you have obtained the proper import authorization.