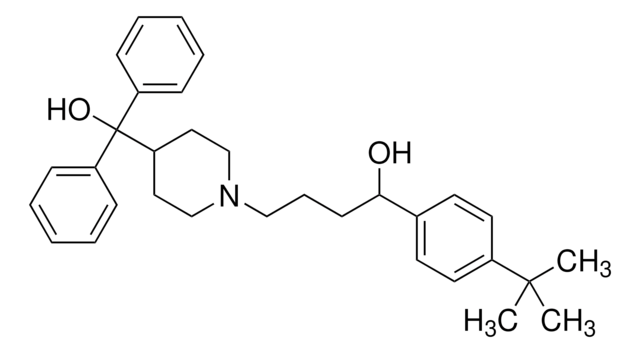
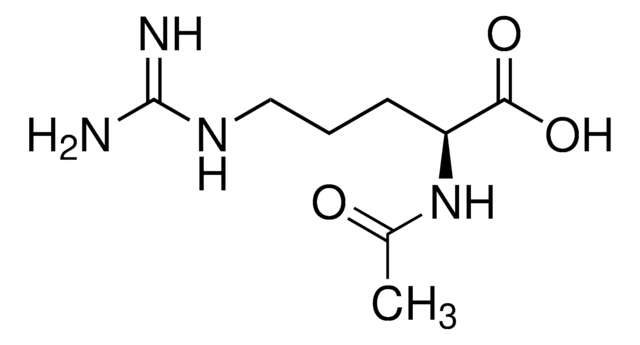
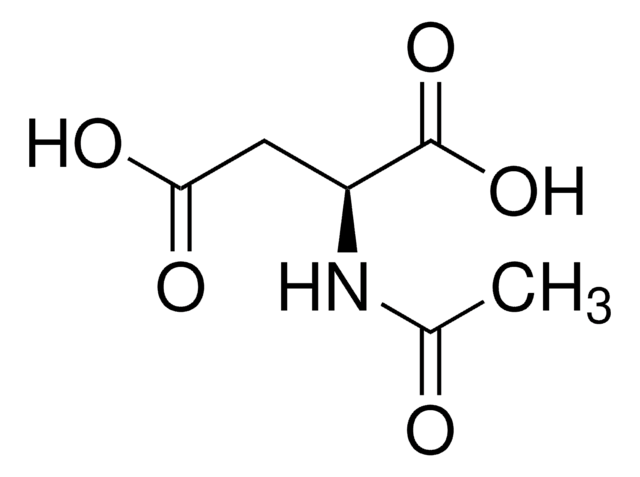
A0783
N-Acetyl-L-proline
≥98% (TLC), suitable for ligand binding assays
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C7H11NO3
CAS Number:
Molecular Weight:
157.17
EC Number:
MDL number:
UNSPSC Code:
12352209
eCl@ss:
32160406
PubChem Substance ID:
NACRES:
NA.26
Recommended Products
Product Name
N-Acetyl-L-proline,
Assay
≥98% (TLC)
Quality Level
form
powder
technique(s)
ligand binding assay: suitable
color
white
storage temp.
2-8°C
SMILES string
CC(=O)N1CCC[C@H]1C(O)=O
InChI
1S/C7H11NO3/c1-5(9)8-4-2-3-6(8)7(10)11/h6H,2-4H2,1H3,(H,10,11)/t6-/m0/s1
InChI key
GNMSLDIYJOSUSW-LURJTMIESA-N
Looking for similar products? Visit Product Comparison Guide
Biochem/physiol Actions
N-Acetyl-L-proline, an analog of the COOH-terminal dipeptide portion of preferred angiotensin-converting enzyme substrates, is use to probe the active site of angiotensin-converting enzyme(s). N-Acetyl-L-proline may be used to to identify, differentiate and characterized N-acyl-amino acid amidohydrolase(s)/aminoacylase(s). N-Acetyl-L-proline is used to study the physicochemical parameters of prolines.
N-acetyl-L-proline is an analog of the COOH-terminal dipeptide portion of preferred substrates of angiotensin-converting enzyme (ACE). It may be used in studies of the binding of substrates and inhibitors by ACE and to differentiate the specificities of various aminoacylases.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Abil E Aliev et al.
The journal of physical chemistry. B, 111(50), 14034-14042 (2007-11-22)
The results of the ring conformational analysis of L-proline, N-acetyl-L-proline, and trans-4-hydroxy-L-proline by NMR combined with calculations using density functional theory (DFT) and molecular dynamics (MD) are reported. Accurate values of 1H-1H J-couplings in water and other solvents have been
J Krapcho et al.
Journal of medicinal chemistry, 31(6), 1148-1160 (1988-06-01)
Analogues of captopril, enalaprilat, and the phosphinic acid [hydroxy(4-phenylbutyl)phosphinyl]acetyl]-L-proline incorporating 4-substituted proline derivatives have been synthesized and evaluated as inhibitors of angiotensin-converting enzyme (ACE) in vitro and in vivo. The 4-substituted prolines, incorporating alkyl, aryl, alkoxy, aryloxy, alkylthio, and arylthio
Mayuko Koreishi et al.
Bioscience, biotechnology, and biochemistry, 69(10), 1914-1922 (2005-10-26)
A novel aminoacylase was purified to homogeneity from culture broth of Streptomyces mobaraensis, as evidenced by SDS-polyacrylamide gel electrophoresis (PAGE). The enzyme was a monomer with an approximate molecular mass of 100 kDa. The purified enzyme was inhibited by the
Chiara Cappelli et al.
The journal of physical chemistry. B, 112(11), 3441-3450 (2008-02-26)
The structure and properties of (s)-N-acetylproline amide (NAP) in aqueous solution are studied by exploiting a continuum solvation model. The conformational preference of NAP as a function of the environment is discussed as well as data for a number of
S N Tenjarla et al.
International journal of pharmaceutics, 192(2), 147-158 (1999-11-24)
A series of N-acetylproline esters (alkyl side chain length, 5-18) were synthesized and tested for potential skin penetration enhancement activity using modified Franz diffusion cells and hairless mouse skin as the penetration barrier. Benazepril and hydrocortisone were used as model
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service