76887
1,4-Dioxane
analytical standard
Synonym(s):
Diethylene oxide, Dioxane
About This Item
Recommended Products
grade
analytical standard
Quality Level
vapor density
3 (vs air)
vapor pressure
27 mmHg ( 20 °C)
40 mmHg ( 25 °C)
Assay
≥99.5% (GC)
autoignition temp.
356 °F
shelf life
limited shelf life, expiry date on the label
expl. lim.
22 %
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
refractive index
n20/D 1.422 (lit.)
n20/D 1.422
bp
100-102 °C (lit.)
mp
10-12 °C (lit.)
density
1.034 g/mL at 25 °C (lit.)
application(s)
cleaning products
cosmetics
environmental
food and beverages
personal care
format
neat
SMILES string
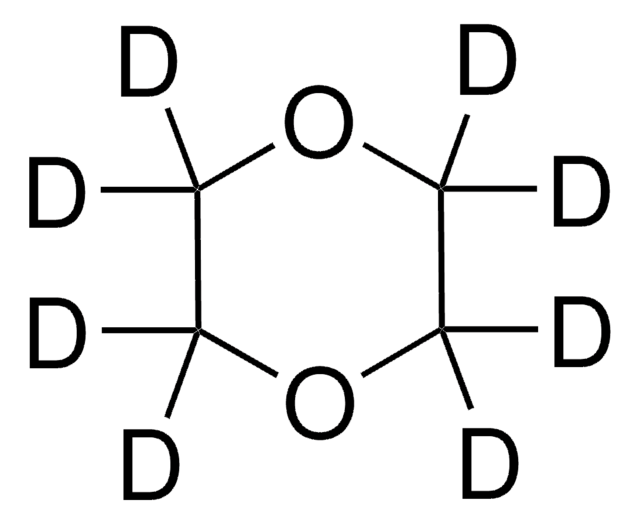
C1COCCO1
InChI
1S/C4H8O2/c1-2-6-4-3-5-1/h1-4H2
InChI key
RYHBNJHYFVUHQT-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Recommended products
recommended
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Carc. 1B - Eye Irrit. 2 - Flam. Liq. 2 - STOT SE 3
Target Organs
Respiratory system
Supplementary Hazards
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
51.8 °F - closed cup
Flash Point(C)
11 °C - closed cup
Personal Protective Equipment
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
EU REACH SVHC Candidate List
EU REACH Annex XVII (Restriction List)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
The Utility of Headspace Grade Solvents for the Analysis of Organic Volatile Impurities
Protocols
US EPA Method 8270 (Appendix IX): GC Analysis of Semivolatiles on Equity®-5 (30 m x 0.25 mm I.D., 0.50 μm)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service