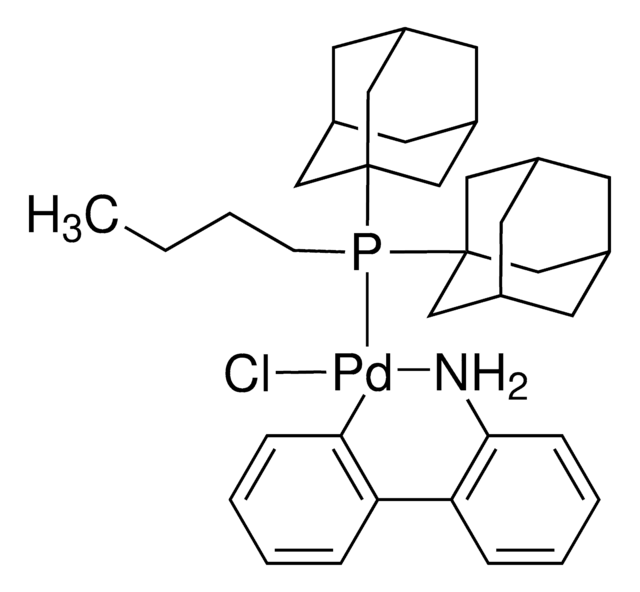
741825
XPhos Pd G2
98%
Synonym(s):
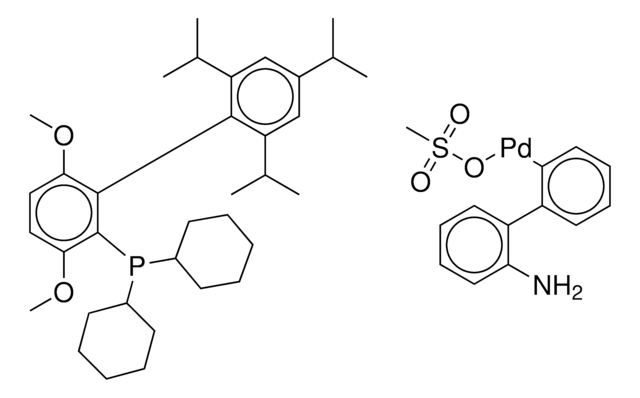
2nd Generation XPhos Precatalyst, Chloro(2-dicyclohexylphosphino-2′,4′,6′-triisopropyl-1,1′-biphenyl)[2-(2′-amino-1,1′-biphenyl)]palladium(II), X-Phos aminobiphenyl palladium chloride precatalyst, XPhos-Pd-G2
About This Item
Recommended Products
Quality Level
Assay
98%
form
solid
feature
generation 2
reaction suitability
core: palladium
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: catalyst
reaction type: Cross Couplings
mp
202-210 °C
functional group
phosphine
SMILES string
Nc1ccccc1-c2ccccc2[Pd]Cl.Nc3ccccc3-c4ccccc4[Pd]Cl.CC(C)c5cc(C(C)C)c(-c6cccc(c6)P(C7CCCCC7)C8CCCCC8)c(c5)C(C)C.CC(C)c9cc(C(C)C)c(c(c9)C(C)C)-c%10ccccc%10P(C%11CCCCC%11)C%12CCCCC%12
InChI
1S/2C33H49P.2C12H10N.2ClH.2Pd/c1-23(2)27-21-31(24(3)4)33(32(22-27)25(5)6)26-14-13-19-30(20-26)34(28-15-9-7-10-16-28)29-17-11-8-12-18-29;1-23(2)26-21-30(24(3)4)33(31(22-26)25(5)6)29-19-13-14-20-32(29)34(27-15-9-7-10-16-27)28-17-11-8-12-18-28;2*13-12-9-5-4-8-11(12)10-6-2-1-3-7-10;;;;/h13-14,19-25,28-29H,7-12,15-18H2,1-6H3;13-14,19-25,27-28H,7-12,15-18H2,1-6H3;2*1-6,8-9H,13H2;2*1H;;/q;;;;;;2*+1/p-2
InChI key
HMRJFNBZAWHTGN-UHFFFAOYSA-L
General description
Application
- Palladium-catalyzed Suzuki-Miyaura coupling reactions of potassium organotrifluoroborates and sulfamates.
- Suzuki-Miyaura cross-coupling reactions of sensitive aryl and heteroarylboronic acids.
- Synthesis of potassium Boc-protected secondary aminomethyltrifluoroborates, via Suzuki-Miyaura cross-coupling reaction.
related product
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
The Heck reaction is the palladium catalyzed cross-coupling reaction between alkenes and aryl or vinyl halides (or triflates) to afford substituted alkenes.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service

![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II), complex with dichloromethane](/deepweb/assets/sigmaaldrich/product/structures/825/986/4317978b-1256-4c82-ab74-6a6a3ef948b1/640/4317978b-1256-4c82-ab74-6a6a3ef948b1.png)